Introduction
Perimenopause, as defined by the North American Menopause Society (NAMS), begins when menstrual cycle variability exceeds seven days and ends one year after the final menstrual period [1]. Although alternative descriptions exist, all characterize it as a transition from predominantly ovulatory cycles to anovulatory cycles, ultimately leading to the cessation of ovulation and permanent amenorrhea. This stage typically starts around age 40, progresses with declining ovarian function, and extends until menopause, which in Western countries occurs at approximately 52 years [2]. In Latin America, the average age at menopause is 48.6 years, according to the Latin American Climacteric Research Network (REDLINC) [3].
During perimenopause, menstrual cycles become irregular, and symptoms such as hot flashes, mood changes, and insomnia may arise, driven by fluctuating estrogen levels. Although fertility declines, ovulation continues intermittently, making effective contraception necessary. This article reviews contraceptive options appropriate for women in perimenopause, supported by scientific evidence regarding their safety and effectiveness [4].
What changes during perimenopause?
Fertility
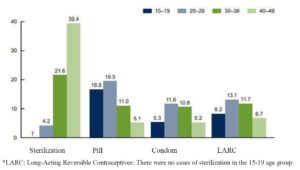
Although fertility begins to decline after age 30 [5], women remain potentially fertile until menopause. Many women underestimate their fertility; an Australian study found that one-third of pregnancies in middle-aged women were unplanned [6]. Pregnancy after age 35 is associated with higher risks, including miscarriage, fetal malformations, gestational diabetes, placenta previa, cesarean delivery, preterm birth, and postpartum hemorrhage [7]. Therefore, effective contraception remains essential during this stage of life. The use of contraceptives decreases with age. Figure 1 illustrates the contraceptive methods used in the United States according to women’s age [8]. However, the World Health Organization (WHO), in its Medical Eligibility Criteria for Contraceptive Use, clearly states that age alone is not a contraindication for any contraceptive method [9].
Menstrual disorders
During perimenopause, over 50% of women experience abnormal uterine bleeding (AUB). The most common patterns include oligomenorrhea or hypomenorrhea (70%), followed by menorrhagia, metrorrhagia, and hypermenorrhea (18%), and amenorrhea (12%) [10]. This irregular bleeding is a major factor leading to hysterectomy, accounting for nearly 60% of cases in the United Kingdom. Hormonal contraceptives help regulate cycles and reduce bleeding volume, providing an effective, less invasive alternative to surgery [11].
Climacteric Symptoms
Approximately 80% of women experience vasomotor symptoms—hot flashes, night sweats, palpitations, and insomnia—during the menopausal transition, significantly affecting quality of life [12]. Salzer had previously described psycho-emotional disturbances in oral contraceptive users over age 40, which were mistakenly ascribed to the contraceptive rather than to the menopausal transition itself [13]. A double-blind study (Table 1) showed that adding estrogens during the pill-free interval markedly reduces hot flashes, headaches, and mood changes [14], underscoring the importance of hormonal fluctuations at this stage.
Studd proposed that abrupt estrogen fluctuations occurring in the postpartum period, the premenstrual phase, and the climacteric transition contribute to psychiatric symptoms such as depression, anxiety, and irritability in susceptible women [15]. In women using cyclic contraceptives who develop climacteric symptoms, the hormone-free interval may further downregulate estrogen receptors, worsening symptoms. Adding estrogen during the pill-free week can prevent this relative hypoestrogenism and improve clinical outcomes [16]. Hormonal contraceptives containing estrogen remain the treatment of choice for severe vasomotor symptoms [17]. Medications such as selective serotonin reuptake inhibitors (SSRIs) may help some women [18], although side effects often limit their use. Alternative therapies have not demonstrated consistent efficacy [19] (Table 2).
Short-acting reversible contraception (SARC)
SARCs are methods that require frequent and regular administration by the user, generally on a daily, weekly, or monthly basis, including:
1. Combined oral contraceptives (COCs)
2. Transdermal patch
3. Vaginal ring
4. Progestin-only pill
Combined oral contraceptives (COCs) in perimenopause
COCs have been in use for more than 50 years, with extensive evidence supporting their efficacy (failure rate of 0.3% with perfect use) and safety. Beyond contraception, they regulate menstrual cycles and represent a major public health achievement [26].
The Royal College of General Practitioners Study, which followed 46,112 women for up to 39 years (378,006 woman-years among never-users and 819,175 among ever-users), demonstrated a significant reduction in overall mortality among COC users (RR 0.88; 95% CI: 0.82–0.93), including lower mortality from cancer, cardiovascular disease, and other causes. The estimated absolute reduction in all-cause mortality was 52 per 100,000 woman-years [27,28].
Current formulations use substantially lower hormone doses. In perimenopause, estradiol-containing COCs are preferred due to their lower estrogenic potency and reduced thrombotic risk. If EE is used, the lowest dose (20 µg) is recommended, although it may increase spotting [29]. A wide range of progestins is available (androgenic, anti-androgenic, glucocorticoid, anti-mineralocorticoid), but no specific progestin has demonstrated superiority for this stage of life.
Regarding dosing regimens, extended-cycle schedules (>21 days) reduce the hormone-free interval, improving bleeding patterns as well as premenstrual and climacteric symptoms, with contraceptive efficacy comparable to the traditional 21/7 regimen. In 2021, a new COC containing estetrol 15 mg and drospirenone 3 mg was approved. Estetrol (E4), a natural fetal estrogen, has shown a favorable hemostatic profile and predictable bleeding pattern, making it a promising option for perimenopausal women [30,31]. A preclinical study demonstrated that estetrol has a neutral effect on breast cancer when used alone or combined with drospirenone or micronized progesterone [32].
Additional benefits of COC use
Ovarian cancer
COCs substantially reduce ovarian cancer risk, with protection increasing as duration of use lengthens. A reanalysis of 45 epidemiological studies including 23,257 cases and 87,303 controls reported a relative risk (RR) of 0.64 (95% CI: 0.59–0.69) after 5–9 years of use and 0.42 (95% CI: 0.36–0.49) after ≥15 years [33]. This benefit persists for more than 30 years after discontinuation. It is estimated that COCs may prevent up to 30,000 ovarian cancer cases worldwide each year.
However, this analysis may be criticized due to its observational design, retrospective data collection, outdated hormone formulations, and exclusion of high-risk women. A subsequent systematic review of 51 studies found a 36% reduction in ovarian cancer incidence among contraceptive users (RR 0.64; 95% CI: 0.60–0.68), with an even greater reduction among COC users (RR 0.62; 95% CI: 0.57–0.68) [34]. Overall evidence quality was rated as moderate.
Endometrial cancer
COCs also protect against endometrial cancer, with longer duration conferring greater benefit. Every 5 years of use is associated with a RR of 0.76, resulting in approximately a 50% reduction after 10–15 years. Protection persists for more than 30 years after discontinuation and does not appear to depend on estrogen dose, parity, body mass index (BMI), or menopausal status [35].
A PROSPERO-registered meta-analysis reported a significant reduction in endometrial cancer risk (OR 0.61; 95% CI: 0.46–0.80). Protection was strongest with ≥10 years of use (OR 0.31; 95% CI: 0.13–0.70), but remained significant at ≥5 years (OR 0.39; 95% CI: 0.23–0.64) and <5 years (OR 0.66; 95% CI: 0.48–0.91) [36]. Notably, 26 of 56 studies (46.4%) were rated as having serious or critical risk of bias due to inadequate control of confounders.
Bone mineral density (BMD)
In women over 40, COCs containing 20 μg EE may help maintain or increase bone mineral density (BMD) [37]. While evidence for reduced fracture risk before menopause remains inconclusive [38], some studies indicate a lower incidence of hip fractures with sustained use.
Among perimenopausal women and those with hypothalamic oligo/amenorrhea [39], COCs have demonstrated a positive impact on BMD. One study comparing three COCs with identical estrogen doses (20 μg EE) but different progestins (desogestrel 0.15 mg, levonorgestrel 0.1 mg, or gestodene 0.75 mg) showed BMD improvement in all groups, with no significant differences between formulations and clear benefit over non-users (p < 0.05) [40].
Risks of COC use
During perimenopause, certain risks must be considered prior to prescribing COCs, including venous thromboembolism (VTE), myocardial infarction, stroke, cervical cancer, and breast cancer. These must be evaluated in the context of individual risk factors and balanced against benefits, which include:
- Effective contraception
- Cycle control and reduced menstrual bleeding volume and pain
- Relief of vasomotor symptoms
- Preservation of BMD
- Protection against ovarian and endometrial cancers
Venous thromboembolism (VTE)
The association between COCs and VTE was first identified in 1961 in a woman using Enovid®, which contained 150 μg mestranol and 10 mg noretynodrel. This triggered extensive surveillance of cardiovascular effects.
Higher-dose EE formulations produce greater alterations in coagulation and fibrinolysis, including increased platelet activation, elevated coagulation factors (II, VII, VIII, IX, X), higher fibrinogen and soluble fibrin, and reduced antithrombin levels [41]. The highest VTE risk generally occurs in the first year of use, likely reflecting previously undiagnosed thrombophilia [42]. According to the UK Faculty of Sexual and Reproductive Healthcare (FSRH), COCs may be safely used until age 50 in healthy, non-smoking women [43].
In recent years, the introduction of 17β-estradiol (E2) or estradiol valerate (E2V) as alternatives to EE has been associated with a lower impact on hemostatic variables, attributable to their faster metabolism to estrone via 17β-hydroxysteroid dehydrogenase and to a more moderate effect on fibrinolysis and endothelial activation [44,45].
Recent data demonstrate that COCs containing estradiol or estradiol valerate are associated with a significantly lower risk of VTE compared with EE-based formulations. In a pooled analysis of two large, prospective, observational cohort studies (INAS-SCORE and INAS-OC), including 11,616 women using estradiol valerate–dienogest and 18,681 using EE–levonorgestrel, contributing 17,932 and 29,140 woman-years of observation, respectively, the adjusted hazard ratio was 0.46 (95% CI: 0.22–0.98), indicating an almost 50% reduction in thromboembolic risk [46].
Another prospective observational study compared the risk of VTE and arterial thromboembolism (ATE) among women from 12 countries in Australia, Europe, and Latin America who used the combined contraceptive estradiol/nomegestrol acetate (NOMAC-E2) versus levonorgestrel-containing combined oral contraceptives (COC-LNG). A total of 101,498 women were followed for up to two years, contributing 144,901 woman-years of observation. The adjusted risk of VTE among NOMAC-E2 users was HR = 0.59 (95% CI: 0.25–1.35), leading to the conclusion that NOMAC-E2 use was not associated with an increased risk of VTE or ATE compared with COC-LNG [47].
Estetrol (E4) is a natural estrogen produced exclusively by the human fetal liver during pregnancy. In 2021, its use in contraception was approved, specifically in combination with 15 mg E4 and 3 mg drospirenone. Available evidence indicates that this new COC exerts a very low impact on the coagulation system. Unlike EE and, to a lesser extent, E2, E4 displays a more neutral hemostatic profile, with minimal effects on coagulation factors. Even when combined with drospirenone, E4 induces significantly smaller changes in procoagulant markers, including factor VII, factor X, fibrinogen, and D-dimer, compared with EE-containing combinations.
E4 is also associated with a more stable coagulation–fibrinolysis balance, as its impact on physiological inhibitors such as antithrombin and protein S is minimal, thereby helping to maintain a more favorable hemostatic profile than EE-based COCs [48].
In phase III clinical trials of E4/drospirenone (Nextstellis®), which included more than 3,500 women followed for up to one year, very few cases of VTE were reported, with no evidence of a significant increase compared with the general population of women of reproductive age [49,50]. Overall, the E4/drospirenone combination appears to offer a safer hemostatic profile than EE and likely a more favorable one than E2. However, long-term follow-up studies are still required to confirm its true impact on VTE incidence in clinical practice.
This scenario highlights the need to update thrombotic risk assessment in oral contraceptive use, differentiating risk according to the type of estrogen employed rather than considering all COCs as a homogeneous group.
The route of administration does not appear to significantly alter this risk. Although it was initially believed that transdermal or vaginal methods might reduce the procoagulant effect by avoiding first-pass hepatic metabolism, studies have shown that both the transdermal patch and the vaginal ring have a thrombotic profile similar to that of oral COCs. This may be due to the direct action of steroids on endothelial cells, which produce procoagulant proteins such as protein S and TFPI [51].
A study comparing a contraceptive patch containing EE and norgestimate with its oral equivalent found no significant differences in the incidence of VTE, reinforcing the evidence that the prothrombotic risk persists regardless of the administration route [52].
Myocardial infarction
In young women, the risk of myocardial infarction is extremely low but increases with age. A meta-analysis including 19 case–control studies and four cohort studies estimated an odds ratio (OR) of 2.5 (95% CI: 1.9–3.2) among users of COCs. The heterogeneity observed across studies indicates that the variation in results is not attributable to chance alone. Sensitivity analyses showed that the adjusted overall OR ranged from 2.30 to 2.75, with the lower limits of the 95% confidence interval consistently above 1 (2.08–2.12) [53].
Importantly, absolute risk estimates from another study suggest that the risk of fatal myocardial infarction is approximately 22 deaths per million women aged 40 to 44 years [54]. The risk of myocardial infarction increases in the presence of smoking, hypertension, diabetes, or dyslipidemia. In healthy, non-smoking women, COCs do not increase the risk of myocardial infarction. COCs containing a 20-µg estrogen dose appear to be safer [55]. Some studies suggest a lower risk with third-generation progestins; however, the available evidence is insufficient to recommend their use in older women [56].
Ischemic stroke
The use of COCs is associated with an increased risk of ischemic stroke. A meta-analysis of 73 studies estimated a relative risk (RR) of 2.75 (95% CI: 2.24–3.38), which decreased to 1.93 (95% CI: 1.35–2.74) with low-dose estrogen formulations. The absolute risk among normotensive women using low-dose COCs is estimated at 4.1 per 100,000 women per year [57].
In addition, the type of progestin influences risk: COCs containing second-generation progestins are associated with a higher risk (RR 2.54; 95% CI: 1.96–3.28) compared with those containing third-generation progestins (RR 2.03; 95% CI: 1.15–3.57) [58]. A cohort study further demonstrated that the risk of ischemic events increases with higher doses of EE (RR 4.5 for 50 µg vs. RR 1.7 for 20 µg). In this study, second-generation progestins were associated with a higher risk (RR 2.2; 95% CI: 1.6–3.0), whereas progestin-only formulations did not significantly increase risk (RR 1.0; 95% CI: 0.3–3.0) [59].
On the other hand, multiple studies have established an association between migraine and stroke risk. A meta-analysis demonstrated that women with migraine have a 2.16-fold increased risk of stroke (95% CI: 1.89–2.48), which rises markedly among COC users with migraine (RR 8.72; 95% CI: 5.05–15.05) [60].
In addition, a meta-analysis including 16 studies found that the use of COCs is associated with an increased risk of ischemic stroke, even with low-dose oestrogen formulations (RR 1.93; 95% CI: 1.35–2.74) [58]. This corresponds to an additional 4.1 ischemic strokes per 100,000 non-smoking, normotensive women using low-oestrogen oral contraceptives per year. No evidence of publication bias was observed.
Breast cancer
The possible association between COC use and breast cancer risk has raised concern, particularly in women over 40 years of age, in whom the baseline incidence of this malignancy begins to rise significantly. Several studies have demonstrated a modest association between current or recent use of hormonal contraceptives and an increased relative risk (RR) of breast cancer. A meta-analysis published in The Lancet reported an overall RR of 1.24 among current or recent users, with a return to baseline risk approximately ten years after discontinuation [61].
These findings have been confirmed by more recent research evaluating modern contraceptives. A prospective cohort study of 1.8 million women in Denmark, followed for more than 10 years, reported an RR of 1.20 (95% CI: 1.14–1.26) among current or recent users of hormonal contraceptives, with no clinically relevant differences between combined and progestin-only formulations [62]. A 2023 UK study reaffirmed these results, reporting RRs between 1.2 and 1.3 across various modern formulations, including those containing desogestrel, gestodene, levonorgestrel, and drospirenone [63].
No clear linear relationship has been observed between the duration of COC use and increased breast cancer risk, nor has it been demonstrated that factors such as age at initiation, family history of breast cancer, or specific progestin type significantly modify the estimated risk [64].
A systematic review and meta-analysis of 74 studies involving 198,579 women comprehensively summarized the evidence on the association between COC use and breast cancer risk by geographic region. The results were as follows: Africa, RR = 1.16 (p = 0.216); America, RR = 1.03 (p = 0.597); Asia, RR = 1.29 (p = 0.014); Europe, RR = 1.01 (p = 0.904); and the Middle East, RR = 1.29 (p = 0.043). This meta-analysis suggests that oral contraceptive use may be associated with an increased risk of breast cancer; however, this association was not statistically significant across all world regions [65].
Although the breast cancer risks associated with COC use, expressed as RR or OR, may appear substantial, the absolute risks are low, particularly in young women. An analysis conducted by the Oxford Cancer Epidemiology Unit estimated that the additional absolute risk of developing breast cancer related to oral hormonal contraceptive use was 8 cases per 100,000 women who used oral contraceptives for five years between the ages of 16 and 20. In women aged 35–39 years, the absolute excess risk increased to 265 cases per 100,000 users [65].
In a preoperative window trial including 30 premenopausal and postmenopausal women with early estrogen receptor–positive (ER+) breast cancer, administration of estetrol (E4) at a dose of 20 mg/day for 14 days induced a significant pro-apoptotic effect in tumor tissue. In contrast, expression of the cell proliferation marker Ki67 remained unchanged in both premenopausal and postmenopausal women [66].
Additionally, in an early-phase study, escalating oral doses of E4 (20, 40, and 60 mg/day for 12 weeks) were evaluated in postmenopausal women with previously treated ER+/HER2− breast cancer at locally advanced and/or metastatic stages. Among the nine patients who completed the treatment period, five demonstrated objective antitumor effects, assessed by computed tomography according to Response Evaluation Criteria in Solid Tumors (RECIST). A complete response was documented in one patient treated with 20 mg/day, while stable disease was observed in one patient in the same dose group and in three patients who received 40 mg/day [67].
Cervical cancer
The risk of cervical cancer increases with oral contraceptive (OC) use beyond five years. The biological mechanism underlying this association is not fully understood, although OCs are presumed to act as a cofactor with human papillomavirus (HPV) infection. Several case–control studies have reported that women who used OCs for 5–9 years had a relative risk (RR) of 2.82 (95% CI: 1.46–5.42) compared with never-users. This risk increased to 4.03 (95% CI: 2.09–8.02) after 10 or more years of use [68]. However, the risk declines after discontinuation of OCs for approximately 10 years. The peak age for cervical cancer in Western countries is between 30 and 35 years. Cervical cancer screening programs have significantly reduced its incidence, particularly among women over 45 years of age.
In a systematic review of 51 studies (two randomized controlled trials and 49 observational studies), pooled data showed a higher risk of cervical cancer among non-users compared with oral contraceptive users (RR 1.28; 95% CI: 1.21–1.35) [34]. These findings were characterized by high heterogeneity.
Transdermal contraceptive
The first transdermal contraceptive approved in the United States and Europe was Evra. It is a 20 cm² patch that releases EE and norelgestromin over a three-week period, followed by a patch-free week. Its contraceptive efficacy is high (Pearl Index: 0.7 pregnancies per 100 woman-years with perfect use), although effectiveness may be reduced in women weighing more than 90 kg. The patch provides cycle control comparable to that of COCs and has a similar side-effect profile, while tending to offer better adherence. This makes it an attractive option for women of all ages, including those in the perimenopausal period [69].
In a study conducted in eight European countries, more than 80% of users reported satisfaction with the patch after three and six cycles. By the end of the study, 73.7% of participants expressed a preference for the patch compared with their previous contraceptive method. Of 4,107 cycles, 3,718 (90.5%) were completed with perfect adherence. Two pregnancies occurred, corresponding to a Pearl Index of 0.63, and the patch was well tolerated [70].
Evidence regarding the risk of VTE associated with the transdermal patch is mixed. Some studies report a thrombotic risk comparable to that of COCs, while others suggest a lower risk. In all cases, the absolute risk of VTE remains very low and is lower than the risk associated with pregnancy [53].
A Cochrane review compared contraceptive effectiveness, cycle control, adherence, and safety of the contraceptive patch or vaginal ring versus COCs. Eighteen trials met the inclusion criteria and were analyzed (six evaluating patches and 12 evaluating vaginal rings). Contraceptive effectiveness was similar across all three methods. Patch users demonstrated better adherence than COC users but also experienced higher discontinuation rates due to adverse events, including breast discomfort, dysmenorrhea, nausea, and vomiting.
Vaginal ring users had higher rates of vaginitis and leucorrhea but reported less vaginal dryness. They also experienced lower rates of nausea, acne, irritability, depression, and emotional lability compared with COC users [71]. The authors rated the overall quality of evidence in this review as low.
Vaginal ring
The vaginal ring is a flexible device measuring 5 cm in diameter that contains EE and etonogestrel, continuously releasing the hormonal combination over a three-week period, followed by a one-week break. It is a convenient and well-accepted method with high contraceptive efficacy. When used in a 21/7 regimen or continuously, it provides better bleeding control compared with COCs. A study conducted in women with type 1 diabetes in the late reproductive stage showed that the vaginal ring does not significantly affect lipid or carbohydrate metabolism or coagulation, suggesting a favorable metabolic safety profile.
Although it has not been specifically studied in perimenopausal women, the vaginal ring is considered a promising method. Its systemic exposure to EE is approximately 50% lower than that of a 30 µg COC, translating into a lower metabolic impact while maintaining contraceptive efficacy, which may make it a safer option for this population group [71–73].
A study evaluated the relationship between use of the contraceptive vaginal ring and the vaginal microbiota using 16S rRNA gene sequencing in 20 women. After two months of treatment, no changes were observed in bacterial quantity or balance, with Lactobacillus crispatus remaining the predominant species. Only one woman with recurrent vulvovaginitis demonstrated increased bacterial diversity with a tendency toward anaerobic bacterial predominance. Overall, the findings indicate that use of the vaginal ring does not negatively affect the composition or structure of the vaginal microbiota [74].
Progestogen only Pills (POP)
Progestogen-only pills (containing levonorgestrel, desogestrel, or norethisterone) are effective and safe options for perimenopausal women. Contraceptive efficacy in older women is high; however, strict daily intake at the same time is required, with the exception of desogestrel-containing pills, which allow a 12-hour dosing window. Bleeding patterns are often irregular but are usually light and painless. The only absolute contraindication to the use of progestogen-only pills is breast cancer [75].
Contraceptive efficacy is maintained through additional progestin-mediated mechanisms. In the case of drospirenone, formulations contain a slightly higher progestin dose than combined contraceptives, contributing to more consistent suppression of ovulation. In a study evaluating a deliberate 24-hour delay in intake to simulate a missed dose, inhibition of ovulation was maintained in nearly all participants, with evidence of ovulation observed in only 1 of 127 women. Additional advantages of progestin-only pills include ease of initiation and discontinuation, rapid return of fertility after cessation—usually by the subsequent cycle—a favorable safety profile, and minimal effects on hemostatic parameters [76].
Long-acting reversible contraceptives (LARC)
These are highly effective, safe methods that do not require daily administration or frequent use.
They include:
1. Long-acting injectables: Medroxyprogesterone acetate (MPA)
2. Subdermal implants
3. Intrauterine devices (IUDs): Copper IUD and levonorgestrel-releasing IUD (hormonal IUD, “intrauterine system”).
Injectable progestogens
Medroxyprogesterone acetate (MPA) is a highly effective contraceptive method administered as an injectable progestin, available in intramuscular (150 mg) and subcutaneous (104 mg) formulations given at 12-week intervals. Its use is associated with adverse effects including irregular bleeding, weight gain, headaches, mood changes, and loss of BMD [77]; therefore, it should be avoided in women with risk factors for osteoporosis. A systematic review evaluating bleeding patterns with MPA (13 studies, 1,610 participants) reported that 46% of users developed amenorrhea within 90 days after the fourth injection [78], increasing to approximately 70% after two years of use [79]. Both contraceptive efficacy and cycle irregularities may persist for up to 12 months following the last injection, likely due to prolonged retention in adipose tissue and sustained suppression of the hypothalamic–pituitary–ovarian axis. The typical-use effectiveness of MPA and progestin-only pills is estimated at 4–7 pregnancies per 100 women per year [80].
Subdermal implants
There are currently two types of subdermal contraceptive implants: levonorgestrel-releasing implants (i.e. Jadelle®, formerly Norplant®) and the etonogestrel-releasing implant (Implanon®/Nexplanon®). Both provide very high contraceptive efficacy (≥99%), long-term protection for 3–5 years, and rapid reversibility after removal [81]. A meta-analysis of 11 studies including 923 women from Europe, Asia, South America, and the United States reported that the bleeding pattern during the first three months of implant use is commonly characterized by prolonged, frequent, or irregular bleeding, which often persists in subsequent months. However, among women experiencing frequent or prolonged bleeding early after insertion, approximately 50% showed improvement within the following three months. Overall, bleeding patterns with contraceptive implants remain unpredictable, and up to 11% of users discontinue the method within the first year, primarily due to irregular bleeding [82].
A potential concern with subdermal contraceptive implants is their effect on BMD, particularly in relation to the risk of osteopenia or osteoporosis. In one prospective study, BMD was assessed at the distal and ultradistal radius of the non-dominant forearm at baseline and at 18 and 36 months after insertion of either an etonogestrel (ENG) or levonorgestrel (LNG) implant [83]. No significant changes in BMD were observed at the ultradistal radius—an area rich in trabecular bone—after 36 months of use with either implant.
Potential pharmacological interactions should be considered. The ENG implant maintains stable serum concentrations when co-administered with isotretinoin, without compromising contraceptive efficacy [84]. In contrast, concomitant use of CYP3A4 enzyme inducers such as topiramate or rifampicin significantly reduces ENG levels, potentially increasing the risk of ovulation and contraceptive failure [85,86].
In the perimenopausal population, implant use may be limited by the need for trained insertion and removal, as well as by side effects such as irregular bleeding, headache, and mood changes, which may reduce acceptability in this age group.
Intrauterine devices (IUD)
Intrauterine devices (IUDs) are among the oldest modern contraceptive methods and are available in two forms: the copper IUD (Cu-IUD) and the levonorgestrel-releasing IUD (LNG-IUD), both of which provide highly effective contraception. The Cu-IUD is also the most effective method of emergency contraception and can be inserted up to 5–7 days after unprotected intercourse. The potential role of the LNG-IUD for emergency contraception is currently under investigation, along with the development of newer devices designed to reduce expulsion and bleeding while improving comfort and duration of use.
LNG-IUDs include Mirena (52 mg, effective for 5–8 years), Kyleena (19.5 mg, 5 years), and Jaydess (13.5 mg, 3 years). All provide contraceptive efficacy exceeding 99%, with amenorrhea rates reaching approximately 20% at 12 months and up to 40% at 24 months.
Use of the LNG-IUD during perimenopause and menopause is safe and offers additional clinical benefits. During perimenopause, effective contraception remains necessary, and many individuals experience heavy or irregular bleeding, which can be effectively managed with an LNG-IUD placed during the menopausal transition. In women with menopausal symptoms who require hormone therapy, a 52 mg LNG-IUD may be used for endometrial protection in place of systemic progestins [87,88]. As the copper IUD provides no hormonal benefit, removal is generally appropriate once menopause has been confirmed.
The LNG-IUD can reduce menstrual blood loss by up to 70%, making it an effective option for the management of AUB. Compared with medroxyprogesterone acetate (MPA) and oral progestins, the LNG-IUD provides superior bleeding control in premenopausal women [89]. It is currently considered the most effective medical treatment for symptoms related to adenomyosis, with minimal systemic side effects and improvements in quality of life comparable to those achieved with hysterectomy [90]. Its therapeutic effect is mediated by decidualization and atrophy of the endometrium, leading to a local hypoestrogenic environment and downregulation of endometrial estrogen receptors due to sustained high intrauterine progestin concentrations. One study demonstrated that treatment with an LNG-IUD achieved outcomes comparable to hysterectomy, as reflected by similar improvements in hemoglobin levels at 6 months and 12 months of follow-up [91].
A Danish nationwide cohort study evaluated the association between use of the levonorgestrel-releasing intrauterine device (LNG-IUD) and the risk of ischemic stroke and intracerebral hemorrhage among women aged 18–49 years. The cohort had a mean age of 30 years and a mean follow-up of 7.1 years. During follow-up, 2,916 women experienced ischemic stroke (24.4 per 100,000 person-years) and 367 experienced intracerebral hemorrhage (3.1 per 1,000,000 person-years).
The incidence rates of ischemic stroke and intracerebral hemorrhage among LNG-IUD users were 19.2 and 3.0 per 100,000 person-years, respectively, compared with 25.2 and 3.1 per 100,000 person-years among non-users. The adjusted incidence rate ratio was 0.78 (95% CI: 0.70–0.88) for ischemic stroke and 0.94 (95% CI: 0.69–1.28) for intracerebral hemorrhage. Thus, use of the LNG-IUD was associated with a 22% lower incidence of ischemic stroke, without an increased risk of intracerebral hemorrhage. These findings raise the hypothesis that levonorgestrel, beyond its contraceptive effect, may confer a protective effect against ischemic stroke [92].
Cardiovascular and oncologic safety of LARC
Progestin-only contraceptives, including oral formulations, injectables, implants, and intrauterine devices, are generally considered safe. An increased incidence of deep vein thrombosis, myocardial infarction, or cerebrovascular events (stroke) has not been demonstrated, with the exception of injectable MPA. In women with multiple cardiovascular risk factors, MPA use has been associated with an increased risk of deep venous thrombosis.
With respect to breast cancer, available data are limited but suggest a risk comparable to that observed with COCs [71,94]. Evidence regarding the impact of progestin-only methods on ovarian or endometrial cancer risk remains insufficient and inconclusive.
BMD loss associated with MPA use has not been clearly linked to an increased risk of fractures. Nevertheless, caution is advised in women with risk factors for compromised bone health, including prolonged corticosteroid use, smoking, thyroid disease, or a family history of osteoporosis. In perimenopausal women, MPA-related reductions in BMD are of particular relevance, as they may contribute to an increased risk of osteoporosis, even though a significant increase in fracture risk has not been demonstrated [10].
Global women’s reproductive health
Most of the data presented in this review originate from the Western Hemisphere and may therefore not fully reflect the global reality. Worldwide, an estimated 270 million women of reproductive age lack access to modern contraceptive methods, corresponding to an unmet need of approximately 9%, with the highest burden observed in Sub-Saharan Africa and other low-income regions [94].
Access to contraception varies substantially across regions. While the prevalence of modern contraceptive use reaches approximately 65% in Southeast Asia, East Asia, and Oceania, it remains markedly lower in Sub-Saharan Africa, where usage is estimated at only 24% [95].
Similarly, access to cancer screening remains highly unequal worldwide. Globally, only 36% of women have ever undergone cervical cancer screening; this proportion increases to 84% in high-income countries but falls below 20% in lower-middle- and low-income settings [96]. Within developing countries, screening coverage varies widely, ranging from 1.1% in Bangladesh to 57.6% in the Democratic Republic of the Congo, with disparities largely driven by socioeconomic status, health system capacity, and geographic location.
Conclusion
Hormonal contraception during perimenopause represents a comprehensive health strategy that extends beyond the prevention of unintended pregnancy. In addition to providing reliable contraceptive efficacy, it offers important non-contraceptive benefits, including regulation of menstrual cycles, reduction of vasomotor symptoms, and potential protection against certain types of cancer, thereby contributing significantly to women’s physical and psychological well-being.
It is essential to recognize that associated risks depend not only on the contraceptive method selected but also on individual patient characteristics, such as age, cardiovascular risk profile, BMD, family history, and metabolic status. Consequently, contraceptive choice should be personalized and evidence-based, with careful consideration of safety, efficacy, and quality-of-life improvement.
Up-to-date, comprehensive, and informed medical counseling is key to maximizing benefits and minimizing risks, enabling women to make well-informed decisions regarding their reproductive health and overall well-being. In this context, hormonal contraception should be viewed not merely as a means of fertility control but as an integral component of healthcare during the later reproductive years and the transition to menopause, supporting a preventive, quality-of-life–oriented approach to healthy aging.
Author contribution
María S. Vallejo conceived the review, drafted, edited and revised the whole document.