Introduction
Endometriosis is a condition in which endometrial glands and stroma develop outside the uterine cavity [1]. It is common, affecting 5–10% of women of reproductive age [2–4], and may appear as peritoneal lesions, endometriomas, or deep infiltrating endometriosis (DIE). Typical symptoms include dysmenorrhea, dyschezia, dyspareunia, dysuria, chronic pelvic pain, and infertility. Beyond physical discomfort, endometriosis can significantly affect psychological health, social functioning, sexual satisfaction, and overall quality of life (QoL) [5–10].
Management includes both pharmacological and surgical options. Treatment should be individualized according to symptom profile, severity, reproductive desire, age, extent and location of disease, and response to previous therapies [11]. Surgery was historically considered the primary treatment. However, concerns over surgical risk, the potential negative impact on ovarian reserve [12–14], the high recurrence rate after surgery [15], and the expanding availability of effective hormonal therapies have shifted practice. For women not seeking conception who present with clinical and imaging findings suggestive of endometriosis, hormonal treatment—particularly combined hormonal contraceptives—has become the preferred first-line strategy for pain control [13]. Despite this shift, evidence guiding the optimal choice of product and regimen for specific lesion types and patient characteristics remains limited.
Treatment response varies widely, from complete symptom resolution to minimal improvement, and symptoms typically recur if therapy is discontinued [16]. Consequently, long-term hormonal treatment is recommended, ideally continuing until pregnancy is desired or menopause occurs [17]. In addition to symptom relief and prevention of recurrence, improving QoL is a fundamental goal.
Nomegestrol acetate 2.5 mg combined with 17β-estradiol 1.5 mg (NOMAC/E₂)—a formulation pairing a strongly antiproliferative progestogen with a natural estrogen of low proliferative activity—has emerged as a promising option. The Spanish Fertility Society (Sociedad Española de Fertilidad) includes NOMAC/E₂ among recommended treatments for patients with endometriosis [18].
The primary objective of this study was to assess whether patients with dysmenorrhea and ultrasound findings suggestive of endometriosis (endometrioma ± deep infiltrating endometriosis [DIE]) experienced improvement in QoL after initiating NOMAC/E₂. Secondary objectives included evaluating changes in the intensity and frequency of dysmenorrhea, noncyclic chronic pelvic pain (CPP), dyschezia, dysuria, and dyspareunia; changes in menstrual bleeding patterns; ultrasound-measured changes in lesion size and DIE; patient satisfaction; and treatment-related side effects.
Methods
This was a prospective cohort pilot study including all patients referred from primary care to our tertiary hospital (Hospital de la Santa Creu i Pau, Barcelona, Spain) with ultrasound findings suggestive of endometriosis who received NOMAC/E₂ between 01/01/2016 and 31/03/2019 for dysmenorrhea management. The study was approved by the Ethics Committee of Hospital de la Santa Creu i Sant Pau (Barcelona, Spain).
Eligible participants were women of reproductive age with regular menstrual cycles (24–38 days), dysmenorrhea ≥ 3 on a visual numeric scale (VNS), and ultrasound findings consistent with endometriosis based on IDEA criteria. The IDEA protocol includes standardized transvaginal assessment of the uterus and adnexa (for adenomyosis and endometriomas), evaluation of ovarian mobility and site-specific tenderness, inspection of the pouch of Douglas for obliteration, and systematic identification of deep endometriosis nodules in the anterior and posterior compartments. Additional inclusion criteria were absence of pregnancy, no hormonal therapy in the preceding 3 months, and no intention to conceive within the next year. Patients with contraindications to combined hormonal contraceptives (WHO eligibility criteria) were excluded. All participants provided written informed consent. Although NOMAC/E₂ is typically administered in a 24/4 regimen, all patients in this study were instructed to use continuous dosing without hormone-free intervals.
During the study period, 187 women were evaluated for suspected endometriosis; 14 met inclusion criteria and started continuous NOMAC/E₂ (Zoely®).
The primary outcome was QoL, assessed using the EuroQol-5D (EQ-5D) questionnaire [19]. The EQ-5D includes a descriptive system covering five domains (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression), each with five severity levels, yielding a global EQ-5D health state score. Patients also rated their overall health using the EQ visual analogue scale (EQ-VAS), a 20-cm vertical scale from 0 (worst imaginable health) to 100 (best imaginable health). EQ-5D scores were calculated using reference values established for our population [20].
Secondary outcomes included duration and intensity of dysmenorrhea, menstrual bleeding patterns, chronic pelvic pain (CPP), dysuria, dyschezia, and dyspareunia. Pain duration was measured in days, and intensity was rated using a VNS ranging from 0 (no pain) to 10 (worst pain imaginable). Patients reported the average number of days with pain, pain intensity, and bleeding days based on the previous three menstrual cycles. Menstrual bleeding intensity was classified using a 4-item Likert scale (amenorrhea, light, normal, abundant).
Ultrasound outcomes included maximum diameter of the largest endometrioma, maximum diameter of the largest DIE lesion, and the sum of maximum diameters of all endometriomas. Need for surgery, treatment changes, adverse effects, and satisfaction were also recorded. Treatment satisfaction was assessed using a VNS from 0 (completely dissatisfied) to 5 (completely satisfied).
Participants attended three visits: baseline (V0), 6-month follow-up (V1), and 12-month follow-up (V2). At each visit, clinical variables were recorded, the EQ-5D was completed, and a transvaginal ultrasound was performed by the same experienced examiner.
To estimate a mean difference of 0.20 units in EQ-VAS with a SD of 0.20 and a precision of 0.10 (95% CI), 13 participants were required; we recruited 14 (p.6, l. 8–15). All variables assessed at each visit were analyzed descriptively. To analyze the changes after starting treatment, the repeat measures general linear model (GLM) was used when the quantitative variables were parametric and the Friedman test when they were nonparametric. Qualitative variables were analyzed with the Fisher or Chi-squared test to assess differences between visits. All data were entered into the study database and analyzed with the SPSS version 26.0 (IBM Corp., Armonk, NY, USA). Differences were considered statistically significant when the p-value was <0.05. The study was approved by the Ethics Committee of Hospital de la Santa Creu i Sant Pau, Barcelona, Spain.
Statistical analysis
Analyses were performed using SPSS version 26.0 (IBM Corp., Armonk, NY, USA). To detect a mean difference of 0.20 units in EQ-VAS with an SD of 0.20 and a precision of 0.10 (95% CI), a minimum sample of 13 participants was required; 14 were included. All variables were analyzed descriptively. Changes across visits were assessed using a repeated-measures general linear model (GLM) for parametric quantitative variables and the Friedman test for nonparametric variables. Qualitative variables were analyzed with Fisher’s exact or Chi-square tests. Statistical significance was set at p < 0.05.
Results
Of the 187 women evaluated for suspected endometriosis, 63 received combined hormonal contraceptives (CHCs) and 51 received a menopausal symptom-guided approach; the remaining women did not start treatment. NOMAC/E₂ was prescribed to the first 14 patients who accepted continuous treatment during at least 12 months.
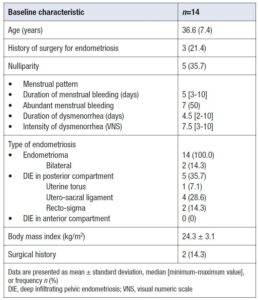
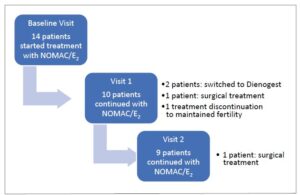
Concomitant conditions such as uterine fibroids >3 cm, adenomyosis, inflammatory bowel disease, and irritable bowel syndrome were excluded during the initial interview. Fourteen patients were included in the final study cohort. The mean (± SD) age was 36.6 ± 7.4 years (Table 1). After the first follow-up visit, 10 patients (71.4%) remained on NOMAC/E₂; after the second visit, 9 (64.3%) continued treatment (Figure 1).
Reasons for treatment modification included insufficient clinical improvement in two cases in which surgery was ultimately chosen. In one patient, the switch to dienogest was motivated by persistent pain (with only slight improvement) and intermenstrual bleeding, whereas in another, mood changes prompted the transition to dienogest.
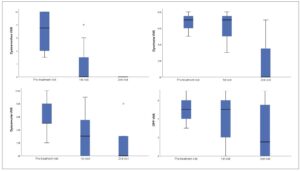
Quality-of-life outcomes demonstrated a significant improvement in both EQ-5D and EQ-VAS scores after treatment initiation (Figure 2). Significant differences were observed between baseline and both follow-up visits. Between the first and second visits, improvement was detected only in the EQ-5D score.
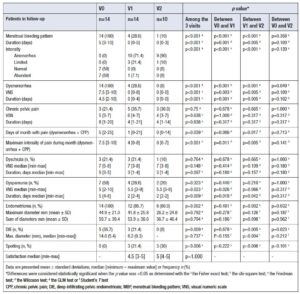
Clinical and ultrasound findings are summarized in Table 2. Patients showed improvements in frequency and intensity of dysmenorrhea, menstrual pain, and menstrual bleeding patterns. However, no reductions were observed in the size of endometriomas or DIE lesions.
No differences in treatment response were associated with body mass index (BMI). With continuous medical therapy, 85.7% of women avoided surgery. Reported adverse effects included spotting (21.4%), decreased libido (7.1%), mood alterations (7.1%), hair loss (7.1%), and a sensation of heaviness in the legs (7.1%). Overall satisfaction with treatment was high, with median VNS scores of 4.5 at the first follow-up and 5 at the second visit.
Discussion
The results of our study suggested that treatment with a continuous regimen of NOMAC/E₂ in women presenting with dysmenorrhea and ultrasound findings suggestive of endometriosis led to a significant improvement in QoL, as measured by the EQ-5D questionnaire, after at least 6 months of uninterrupted therapy. Specifically, both the EQ-5D index score and the EQ-VAS showed statistically significant increases, indicating a marked enhancement in patients' perceived overall health status and well-being.
Endometriosis significantly impairs quality of life (QoL), as women with the condition consistently report lower QoL compared to those without the disease [5-10]. CCP, dysmenorrhea, dyspareunia, and associated physical and psychological symptoms contribute to social isolation, anxiety, depression, and reduced sexual satisfaction, collectively diminishing overall well-being [11]. Epidemiological evidence indicates that 60–70% of affected women experience physical, psychological, or social impairments that substantially affect their QoL [11-13].
Management strategies that address both symptom relief and holistic well-being are essential. Combined oral contraceptives such as NOMAC/E₂ have shown potential in alleviating pain and improving QoL, providing meaningful benefits in this population. Perceived QoL is also influenced by factors such as disease acceptance, pain severity, BMI, intimate relationships, financial burden, age, socioeconomic status, and educational level [14], emphasizing the importance of individualized treatment. Severity and duration of symptoms, as well as diagnostic delays, correlate with further reductions in QoL [7,15,13], highlighting the need for early recognition and effective management. Routine assessment using validated QoL tools such as SF-36, EHP-30, EHP-5, and EQ-5D [21,22] is crucial for evaluating treatment efficacy. Despite its relevance, research specifically analyzing QoL outcomes in endometriosis remains limited [23], underscoring the need for continued investigation into treatments that optimize both pain control and overall life quality.
In recent years, several studies confirmed that different combined oral contraceptive regimens are safe and associated with improved symptoms and reduction in endometriosis lesions [24-26].
Within broader CHC evidence, randomized and prospective data indicate that extended or continuous regimens reduce endometriosis-related pelvic pain and improve health-related QoL. Flexible/extended ethinyl-estradiol/drospirenone significantly lessens pelvic pain versus placebo and supports QoL gains in randomized trials [27]. Comparative studies of dienogest/ethinyl estradiol show that continuous regimens yield greater pain reduction and better QoL than conventional cyclic dosing [28]. For estradiol-based CHCs, the NOMAC/E₂ pill improves CCP and QoL in observational and randomized settings, and head-to-head data show comparable pain relief to dienogest monotherapy, with some time-point advantages for dienogest on somatic QoL domains [29]. Evidence for estradiol valerate/dienogest is more limited, but available reviews describe improvements in dysmenorrhea and QoL similar to other CHCs [30].
Collectively, current literature supports CHCs—particularly in extended/continuous regimens—as effective options for alleviating dysmenorrhea and pelvic pain while enhancing QoL; however, no regimen demonstrates clear, consistent superiority across outcomes, underscoring the need for individualized choices based on symptoms, tolerability, preferences, and comorbidities. Škegro et al. showed that higher pain levels correlated with poorer QoL [31]. Studies using drospirenone/EE demonstrated reductions of up to 40% in endometrioma size [32,33]. Scala et al. [34] evaluated levonorgestrel/EE in an 84/7 regimen for 12 months and observed significant reductions in symptoms and lesion size. Xholli et al. [35] reported significant reductions in endometrioma size and disappearance of lesions in nearly half of women treated with dienogest/EE for at least 12 months, along with decreases in dysmenorrhea, intermenstrual pain, and dyspareunia. Yoshino et al. [36] observed QoL improvement in Japanese women with dysmenorrhea or endometriosis-associated pain treated with extended drospirenone/EE. QoL improvement has also been reported in women who switched from cyclic to extended regimens [37].
Regarding strengths, this prospective cohort pilot design allowed temporal evaluation of continuous NOMAC/E₂ therapy on QoL and clinical outcomes in women with ultrasound-suspected endometriosis. The validated EQ-5D instrument enabled multidimensional, patient-centered assessment, capturing changes in mobility, self-care, daily activities, pain/discomfort, and anxiety/depression. Another strength was consistency in clinical and ultrasound evaluations, all performed by a single experienced operator, minimizing inter-observer variability. The longitudinal follow-up at baseline, 6 months, and 12 months allowed assessment of both short- and intermediate-term effects.
Nonetheless, several limitations must be acknowledged. The small sample size (n = 14) restricts statistical power and limits generalizability. The absence of a control group prevents definitive attribution of improvements to NOMAC/E₂, as changes may reflect placebo effects or natural disease variation. As a single-center study, selection bias cannot be excluded. Although follow-up extended to 12 months, this period may be insufficient to determine long-term sustainability. Attrition was notable, with up to 40% discontinuation, which may introduce bias. Diagnosis was based on clinical and ultrasound findings rather than histological confirmation, raising the possibility of misclassification.
Conclusions
Treatment with NOMAC/E₂ in women with dysmenorrhea and ultrasound suspicion of endometriosis may significantly improve QoL as measured by EQ-5D. These findings support NOMAC/E₂ as a promising therapeutic option, although further studies are needed to confirm its benefits relative to other treatments. In our study, we did not observe reductions in lesion size, likely due to the small sample and relatively short follow-up. This highlights the importance of prioritizing patient-reported outcomes such as QoL and symptom relief, rather than relying solely on anatomical changes. Future research should include randomized controlled trials with larger cohorts and longer follow-up to confirm these results and compare different hormonal regimens.
Author contributions
Josep Perelló-Capó, Noelia Rams, Rocío Luna, Josep Estadella, Taisiia Melnychuk and Ramon Rovira conceived the study, contributed to clinical data and the main research concept.
Josep Perelló-Capó, Bruna Miarons and Natalia Teixeira: drafted the manuscript, conducted statistical analyses and performed the literature review.
Josep Perelló-Capó, Bruna Miarons, Natalia Teixeira and Rocío Luna: supported manuscript preparation, contributed additional data and participated in supervision.
Josep Perelló-Capó, Noelia Rams, Rocío Luna, Josep Estadella, Taisiia Melnychuk, Ramon Rovira: collected clinical data and reviewed the manuscript.
Josep Perelló-Capó, Noelia Rams, Josep Estadella and Taisiia Melnychuk supervised the study and provided critical revisions.
Josep Perelló-Capó, Rocío Luna and Ramon Rovira contributed with senior scientific input to the research design and the manuscript preparation, and additional statistical analysis.
All authors reviewed the manuscript and approved the final version.
Data availability statement
The data that support our finding are available from the corresponding author (Josep Perelló Capó) upon reasonable request.