Introduction
Preterm birth (PTB), defined as delivery before 37 weeks of gestation, remains the leading cause of neonatal mortality worldwide and a major contributor to long-term morbidity. It affects approximately 15 million births annually [1], accounting for nearly 11% of all live births, or roughly one in every ten babies born globally [2,3]. Complications related to PTB cause more than one million neonatal deaths each year, many of which could be prevented through timely, evidence-based interventions.
There is marked geographical variation in PTB rates, with the highest burdens observed in South Asia and sub-Saharan Africa, where rates often exceed 12–14% [4]. In contrast, several high-income countries, particularly in the Nordic and Baltic regions, report some of the lowest PTB rates worldwide [5]. According to the EURO-PERISTAT report, PTB rates across Europe vary widely, ranging from below 6% in countries such as Latvia, Estonia, Finland, and Lithuania to above 8% in Belgium, Germany, and Greece. Latvia’s stable PTB rate of 5.45% places it among the lowest in Europe and closely aligns with its Baltic neighbors, providing important regional context for interpreting national outcomes [6].
This achievement raises an important question: can lessons from countries with consistently low PTB rates be applied to higher-risk settings? Understanding the biological mechanisms and health system features that sustain this stability may help inform strategies to reduce PTB globally, particularly through strengthening health systems and enhancing maternal and newborn care.
This article is presented as a narrative review that integrates epidemiological data from Latvia with expert clinical insights. While it is not intended as a systematic review, it provides a clinically focused synthesis of available evidence and practical experience.
The concept of the “Great Obstetrical Syndrome,” proposed by Romero et al. [7], offers a useful framework for understanding the multifactorial causes of preterm birth (PTB) and other adverse pregnancy outcomes. Rather than arising from a single pathological process, PTB reflects the convergence of multiple interconnected biological pathways.
At the core of this model is the recognition that both spontaneous and medically indicated preterm births may arise from distinct mechanisms, including, but not limited to, cervical insufficiency, intrauterine infection or inflammation, uterine overdistension, placental insufficiency, decidual senescence, and breakdown of maternal–fetal immune tolerance. These mechanisms frequently overlap and may act synergistically, culminating in the premature activation of parturition pathways.
Furthermore, the model underscores the importance of maternal, fetal, and placental characteristics, along with broader determinants such as socioeconomic status, nutritional status, psychosocial stress, and access to antenatal care. Maternal gynecologic history (i.e. prior cervical surgery), comorbidities (i.e. diabetes and hypertensive disorders), and environmental exposures also play crucial roles in shaping individual risk profiles.
By framing PTB within a systems-based perspective, the Great Obstetrical Syndrome emphasizes the importance of personalized care and targeted interventions. This approach supports a shift from universal strategies toward risk-based methods, particularly for high-risk groups such as women with a short cervix or suspected cervical insufficiency.
Latvia’s trends in preterm birth
Over the past two decades, Latvia’s average preterm birth (PTB) rate has remained stable at 5.45%, although notable shifts have occurred in its composition and associated outcomes. The most prominent trend has been a decline in spontaneous PTB among singleton pregnancies, particularly in very and extremely preterm births [6]. In parallel, there has been a moderate increase in medically indicated late preterm births, likely reflecting changes in clinical decision-making and obstetric practice [8,9]. These structural shifts have been accompanied by a substantial reduction in perinatal and neonatal mortality, especially among births managed at tertiary-level perinatal centers.
Multiple gestation remains the strongest risk factor for PTB, contributing disproportionately to overall rates and particularly to early gestational age subgroups. Other major contributors include a history of prior preterm delivery, advanced maternal age, and chronic maternal comorbidities such as obesity, hypertension, and diabetes. Notably, the increasing use of assisted reproductive technologies and a rise in elective deliveries have influenced the epidemiological landscape, contributing to the observed increase in late iatrogenic PTB. At the same time, favorable sociodemographic trends may have supported the decline in spontaneous PTB, including a steady reduction in maternal smoking prevalence and gradual improvements in educational attainment among women of reproductive age [10]. These factors may have promoted healthier pregnancy behaviors and earlier engagement with antenatal care.
Importantly, the Latvian experience suggests that stability in national PTB rates can obscure meaningful improvements in neonatal outcomes when accompanied by targeted preventive strategies and system-level enhancements. In this context, the reduction in spontaneous very and extremely preterm births, together with perinatal system reorganization, appears central to the observed gains in neonatal survival.
Screening strategies for identifying women at risk of preterm birth: a stepwise approach toward individualized prevention
Early identification of women at increased risk of preterm birth (PTB) is essential for effective prevention. A prior episode of spontaneous or medically indicated PTB remains one of the strongest and most reliable predictors of recurrence, conferring up to a twentyfold increased risk [11]. Accordingly, risk assessment before conception and the collection of a detailed antenatal history early in pregnancy are critical for optimizing surveillance, initiating timely interventions, and providing individualized counseling.
Transvaginal ultrasound
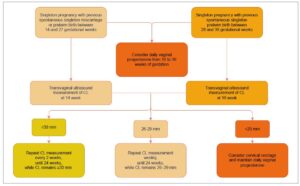
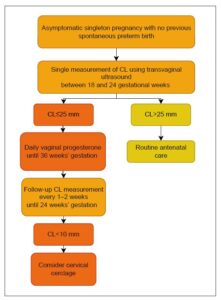
In line with the ISUOG Practice Guidelines on the use of ultrasound for predicting spontaneous preterm birth, Latvia has implemented routine cervical length (CL) measurement during the second-trimester anomaly scan for all pregnant women [8]. Transvaginal ultrasound performed between 18 and 24 weeks of gestation—supported by high-level evidence and a grade A recommendation for women with a history of PTB—enables early detection of a short cervix (≤25 mm) [12], a significant and modifiable risk factor for spontaneous preterm birth in women both with and without a prior history of PTB [13].
This universal screening strategy facilitates timely and targeted preventive interventions, including vaginal progesterone therapy (moderate- to high-quality evidence; grade A recommendation for women with a history of PTB and grade C for those without a prior PTB; Figure 1) [14] and cervical cerclage in selected high-risk women (moderate- to high-quality evidence; grade C recommendation) [15]. At the same time, it helps minimize unnecessary interventions in low-risk pregnancies (Figure 2).
Biochemical markers
Biochemical markers in cervicovaginal secretions and maternal blood have been extensively studied to improve the early detection of preterm birth (PTB), particularly in symptomatic women. Fetal fibronectin (fFN), a glycoprotein, is among the most well-validated tools and serves as a strong negative predictor of delivery within 7–14 days. Other cervicovaginal biomarkers, such as phosphorylated IGFBP-1 (phIGFBP-1) and placental alpha microglobulin-1 (PAMG-1), have demonstrated comparable short-term predictive performance in meta-analyses, although they are less frequently used in routine clinical practice [16].
Inflammation-related markers, particularly interleukins (IL-6, IL-8) and matrix metalloproteinase-9 (MMP-9), have shown consistent associations with PTB, especially in the presence of infection [17]. However, their predictive accuracy remains moderate and highly context dependent. More recently, attention has shifted toward multi-marker panels incorporating proteomics, metabolomics, and lipidomics, often supported by machine learning approaches [18–20]. To date, only a limited number of these panels have been validated for routine clinical use.
Integrative timing and risk stratification
The timing and integration of these risk assessment methods follow a tiered approach, beginning preconceptionally with obstetric history, progressing to structural assessment through cervical length measurement in the second trimester, and incorporating biochemical markers later in pregnancy when clinically indicated. This strategy seeks to direct interventions toward women most likely to benefit, thereby reducing overmedicalization, unnecessary anxiety, and unwarranted procedures in low-risk populations. It also aligns with the principles of personalized and precision obstetric care.
From short cervix to cervical insufficiency
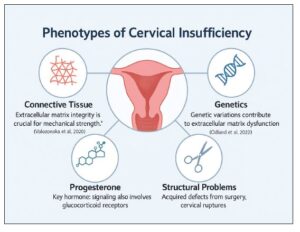
While a short cervix identified on transvaginal ultrasound is a well-established predictor of spontaneous preterm birth, it represents a common pathway rather than a single clinical entity. A phenotype-based approach allows differentiation between functional cervical shortening—often dynamic, reversible, or responsive to progesterone—and true cervical insufficiency, which is characterized by a structurally or biologically compromised cervix incapable of sustaining pregnancy.
In many women, this phenotype results from degradation of the extracellular matrix (ECM), potentially driven by subclinical infection, sterile inflammation, or dysregulated progesterone signaling, all of which weaken cervical collagen structure and biomechanical integrity. In others, the underlying cause is anatomical trauma, such as prior cervical conization, repeated dilation and curettage, or excessive mechanical stress during previous deliveries. These mechanisms are not mutually exclusive and may coexist, particularly in women with recurrent or early-onset cervical shortening.
By integrating ultrasound findings with clinical history, inflammatory biomarkers, and molecular profiling, clinicians can more accurately distinguish between phenotypes that may benefit from expectant management, progesterone therapy, cervical cerclage, or targeted anti-inflammatory interventions. This shift from length-based thresholds to mechanism-based classification supports a more precise and personalized strategy for preventing preterm birth in women with a short cervix.
Genetic insights of preterm birth and cervical insufficiency
Cervical insufficiency (CI) is a significant contributor to second-trimester pregnancy loss and severe preterm birth; however, its underlying etiology remains unclear in many cases. In a study by Volozonoka et al., 21 women with isolated CI were evaluated using next-generation sequencing, revealing rare variants in 12 collagen-related genes. Notably, 67% of these women carried variants in extracellular matrix (ECM) genes commonly associated with connective tissue disorders, such as Ehlers–Danlos syndrome. These findings suggest that CI may represent a mild or subclinical form of collagenopathy rather than a classic monogenic disorder. The high rate of preterm birth observed in this cohort (48.6%) underscores the clinical relevance of these genetic alterations and supports consideration of genetic screening in the diagnostic evaluation of CI, particularly in recurrent or high-risk cases, to inform early interventions such as cervical cerclage [21].
A subsequent genomic study [22] involving 114 women with a short cervix or isolated CI reported that 67% carried rare variants in collagen or ECM-related genes. Four women (3.5%) harbored pathogenic or likely pathogenic (P/LP) variants, predominantly affecting collagen types I, III, and V. Although no single gene accounted for the condition, the cumulative variant burden supports a polygenic and multifactorial etiology. Importantly, this study also identified an enrichment of rare damaging variants in genes involved in progesterone signaling, including PGR (n=8), NR3C1 (n=6), and HSP90AA1 (n=6). These genes play key roles in hormonal responsiveness, stress adaptation, and receptor stability during pregnancy. The coexistence of variants in both ECM and hormonal pathway genes suggests a mechanistic interaction influencing cervical remodeling. Women carrying P/LP variants experienced a significantly higher rate of preterm birth (60%) compared with non-carriers (24%), highlighting the potential clinical value of genetic screening for risk stratification and personalized management. Collectively, these findings support the inclusion of connective tissue and hormonal gene panels in the diagnostic workup of recurrent or early-onset CI, with implications for interventions such as cerclage, progesterone therapy, and intensified surveillance (Figure 3).
The analysis also revealed a significant excess of damaging variants in genes involved in steroid hormone signaling, particularly PGR, NR3C1, and HSP90AA1, which may help explain why some women fail to respond to progesterone therapy. For example, HSP90AA1 encodes a chaperone protein essential for stabilizing steroid hormone receptors during pregnancy; dysfunction in this pathway may contribute to premature cervical remodeling.
From microbiota to molecular markers: new horizons
Dysbiosis of the vaginal microbiota is increasingly recognized as a key contributor to preterm birth (PTB), particularly through pathways involving inflammation and immune activation [23,24]. The reviewed studies consistently report that a reduction in protective Lactobacillus species—especially L. crispatus, L. gasseri, and L. jensenii—together with an overrepresentation of L. iners, Gardnerella vaginalis, Ureaplasma spp., and Prevotella spp., is significantly associated with an increased risk of PTB. This shift in microbial composition, often characterized by an elevated vaginal pH (>4.5) and increased levels of inflammatory cytokines such as IL-1β and IL-6, may contribute to cervical remodeling, membrane weakening, and the initiation of preterm labor.
In addition, the predominance of L. iners, which lacks the protective production of D-lactic acid and hydrogen peroxide, may compromise vaginal acidity and innate host defenses, thereby creating a permissive environment for inflammation and ascending infection. Importantly, emerging evidence suggests that these microbial patterns are influenced by ethnicity, socioeconomic factors, and behaviors such as vaginal douching, underscoring the need for personalized approaches to microbiota monitoring and intervention [25].
Clinical management and timing of birth
Vaginal progesterone is a cornerstone therapy for preventing preterm birth (PTB) in women with a short cervix, particularly in singleton pregnancies. Randomized trials demonstrate significant reductions in PTB before <33, <34, and <37 weeks, accompanied by improved neonatal outcomes, including fewer neonatal intensive care unit admissions and reduced respiratory morbidity [14,26]. When cervical length is markedly shortened—especially <15 mm—or when there is a history of spontaneous PTB, cervical cerclage offers additional benefit. In singleton pregnancies with a prior PTB and a cervical length <25 mm (most pronounced below 15 mm), ultrasound-indicated cerclage has been shown to significantly reduce recurrent PTB and improve neonatal outcomes [15,27].
Emerging evidence also supports targeted antibiotic therapy in selected women with cervical insufficiency and evidence of intra-amniotic infection or sterile inflammation. In a cohort of 22 pregnancies with painless cervical dilation >1 cm and confirmed intra-amniotic inflammation or infection, a triple-antibiotic regimen resulted in resolution of inflammatory markers in 75% of cases, with an overall treatment success rate of 59%, defined as resolution of inflammation or delivery at ≥34 weeks. Notably, pathogenic microorganisms were identified in only 14% of cases, underscoring the predominance of sterile inflammation [28]. Similarly, among 50 women with preterm labor and intact membranes, antibiotic treatment resolved intra-amniotic inflammation in 79% of cases, with 84% achieving a favorable outcome [29]. Together, these findings suggest that antibiotics may modulate both microbial and sterile inflammatory pathways, thereby prolonging gestation and improving perinatal outcomes.
However, as these data derive primarily from small or single-center cohorts, their generalizability remains limited. Broader application should be approached cautiously until validated by larger, multicenter studies.
Our retrospective review conducted at Riga Maternity Hospital between 2019 and 2024 included 154 women with singleton pregnancies diagnosed with cervical insufficiency. Participants were stratified into a cerclage group (n = 107) and a non-cerclage group (n = 47), the latter comprising 45 symptomatic women (unpublished data). No fetal deaths were recorded in the cerclage group, whereas the non-cerclage group experienced a significantly higher risk of adverse outcomes. Across both groups, antibiotic therapy was associated with a reduced incidence of preterm birth before 28 weeks and a lower rate of preterm prelabor rupture of membranes (PPROM), underscoring the clinical relevance of infection and inflammation management [30].
The clinical management of women with a short cervix or cervical insufficiency remains challenging due to the multifactorial nature of the condition. In real-world practice, microbial infection and sterile intra-amniotic inflammation frequently coexist, complicating both diagnosis and therapeutic decision-making. Moreover, it is often difficult to determine whether inflammation is the primary driver of cervical remodeling or a secondary consequence of mechanical cervical failure. These uncertainties reinforce the need for individualized, phenotype-based management strategies that integrate structural, inflammatory, and clinical risk profiles.
When preterm delivery is unavoidable, it is essential to follow evidence-based practices to improve neonatal outcomes, especially in countries with low fertility and declining birth rates, where Every Baby Counts. Early recognition of imminent preterm birth allows for timely interventions such as administering antenatal corticosteroids and magnesium sulphate for neuroprotection, and transferring the mother to a high-level perinatal care center [31,32]. Delivery at experienced perinatal centers significantly reduces neonatal mortality for births before 32 weeks, thanks to immediate access to neonatal specialists, advanced equipment, and coordinated care [33,34]. Centralization of very preterm deliveries to hospitals with the highest level of neonatal expertise is strongly associated with better survival and fewer complications [35,36]. Integrating preterm outcomes into national monitoring systems is essential to ensure consistent quality, drive improvement, and guide evidence-based resource allocation. Success also depends on selecting the safest delivery method. Cesarean section reduces perinatal mortality in preterm births by about 25%, particularly in breech presentations, where the relative risk of adverse outcomes is halved compared to vaginal delivery. In contrast, no significant benefit is observed for cephalic presentations. Neurologically, cesarean delivery is associated with lower morbidity overall, though male neonates remain at higher risk than females regardless of delivery mode [37,38]. Physiological deferred cord clamping [39] and effective respiratory support [40] are crucial components of the preterm infant’s “golden hour.”
The call to action is clear: Europe must develop unified standards for managing preterm birth, ensure equitable access to advanced perinatal care, and implement robust monitoring systems to track outcomes and drive continuous improvement.
Conclusion
Preterm birth continues to be a complex challenge that needs both broad system strategies and tailored interventions. Combining genetic research, microbiota studies, and evidence-based practices paves the way for more precise, phenotype-focused treatments. Each preterm birth presents a chance to implement personalized care, as every infant’s life is valuable.