Introduction
Combined oral contraceptives (COCs), composed of estrogen and progestin, have been widely used since the 1960s. Initially formulated with ethinyl estradiol (EE), efforts aimed to reduce venous thromboembolism (VTE) risk by lowering the EE dose, while in parallel new progestins were developed to improve tolerability [1]. Nevertheless, these newer progestins, once combined with EE even at lower doses, presented a higher VTE risk compared to EE associated with levonorgestrel.
Thus, despite several strategies implemented to mitigate risk, VTE associated with COCs remains a significant public health concern, with an estimated incidence of 5 to 12 cases per 10,000 woman-years depending on the COC used [1]. In Europe and the United States, more than 22,000 and 9,000 COC-associated VTE cases occur each year, respectively [1,2]. Due to concerns about VTE risk, regulatory authorities such as the EMA and the FDA required a re-assessment of COCs about a decade ago. Currently, combinations such as EE/levonorgestrel (EE/LNG) and EE/norgestimate (EE/NGM) are considered preferred options in several countries because of their well-documented thrombotic risk profiles [3]. Based on this information and a comprehensive review of existing data, the EMA concluded in 2014 that the benefit–risk balance of COCs remained favorable [4,5]. However, improving their safety profile still requires new strategies.
Since the late 2000s, a new generation of COCs containing body-identical estrogens (i.e., estradiol (E2) and estetrol (E4)) has been introduced, further supporting the role of the estrogen component in modulating VTE risk. Converging evidence from biological studies [6,7], observational cohorts (e.g., INAS-SCORE, PRO-E2) [8-10], and real-world pharmacovigilance data [11] suggests that these body-identical estrogens are associated with a lower VTE risk compared to EE-based combinations. In 2024, a meta-analysis by Douxfils et al. [10], combining five observational studies, reported a 33% reduction in VTE risk with body-identical estrogens compared to EE-based COCs. As new epidemiological data have been published since 2024, it is timely to update this meta-analysis and to consider body-identical estrogens as the reference therapy when discussing VTE risk.
This article therefore aims to update the meta-analysis of Douxfils et al. [10] by incorporating the most recent observational studies and strengthening current evidence regarding the safer thrombotic profile of COCs containing body-identical estrogens. The goal is to highlight the higher VTE risk associated with EE-based COCs compared to those containing body-identical estrogens.
Methods
This systematic review and meta-analysis was conducted in accordance with the 2020 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement as well as PRISMA-Search for literature searches [12]. The complete methodology has been described elsewhere [10]. An updated protocol for this review and meta-analysis was registered in the Open Science Framework (OSF, ID https://osf.io/n9dav/).
The electronic databases Medline (via Ovid) and Embase were searched in June 2025 for new observational studies reporting the risk of venous thromboembolism (VTE) associated with synthetic estrogens versus body-identical estrogens. This update followed the same search strategy as the previous meta-analysis [10]. The full search strategy for both databases is available in the OSF (ID https://osf.io/n9dav/). In addition, reference lists of relevant articles were manually screened, and experts in the field were contacted to identify any missing studies.
Search results from both electronic and manual screening were imported into Covidence for data management. Covidence is a web-based platform designed to streamline the production of systematic and other literature reviews. Three independent reviewers (LR, JD, CB) conducted title/abstract and full-text screening to identify longitudinal observational studies evaluating VTE risk in women using synthetic estrogens versus body-identical estrogens. Each reference was assessed by up to two reviewers, with discrepancies resolved through discussion and consensus. An Excel extraction form was used to summarize all included studies. Data were extracted by one reviewer (LR) and checked by another (CB). Study quality was assessed using the Newcastle–Ottawa Scale (NOS) for non-randomized studies, independently by two reviewers (LR, CB).
In the meta-analysis, EE-based COCs (i.e., COCs containing EE with any progestogen) or EE/LNG were compared with body-identical estrogen–based COCs (E2-based COCs). All E2-based COCs (E2/nomegestrol acetate (NOMAC), E2/dienogest (DNG)) were pooled. The number of VTE events in each study was combined using a random-effects meta-analysis, as heterogeneity between studies was expected a priori. Peto odds ratios and 95% confidence intervals (CIs) were reported to account for the low incidence of events, as recommended in the Cochrane Handbook for Systematic Reviews of Interventions [13]. For EE/LNG comparisons, when adjusted hazard ratios (HRs) were provided by study authors, pooled HRs were estimated using a random-effects model.
To confirm model robustness, a second adjusted analysis was performed that included only the post-authorization safety studies (PASS) required by the EMA, which assess the safety and benefit–risk profiles of E2/NOMAC and E2/DNG [8,9]. These studies share similar designs and were approved by the EMA. Only confirmed VTE events were included. To allow comparison across studies, VTE events were standardized as events per person-time for prospective cohorts or events per number of participants for case-control studies.
Two sensitivity analyses were performed: a leave-one-out analysis and a subgroup analysis by study design (cohort or case-control). Because of the small number of included studies (k < 10), no publication bias assessment was conducted. For all meta-analyzed associations, the level of evidence was evaluated using the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach [14].
Results
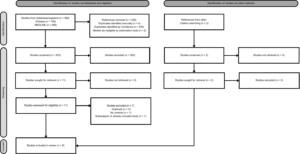
The updated search strategy retrieved a total of 78 additional records: 12 from Medline and 66 from Embase (Figure 1). None of these records met the inclusion criteria. However, the updated manual search identified one additional reference, a research letter containing original data [15]. This reference provided the most recent findings from the Danish population-based cohort, extracted from the Danish National Registries. As a result, Lidegaard et al. [16] from the previous meta-analysis was replaced with Yonis et al. [15]. Although both studies were based on Danish national registry data, Lidegaard et al. included women from January 2009 through December 2011, while Yonis et al. included women followed from January 2011 through July 2021. Therefore, this updated meta-analysis includes five studies [8,9,15,17,18].
Study characteristics
Characteristics of the included studies are presented in Table 1. Studies were published between 2021 and 2025 and included 2,343,585 women/time from cohort studies and 8,514 women from case-control studies. Three cohort studies and two case-control studies were included. All studies demonstrated excellent methodological quality according to the NOS scale: four studies were rated 9/9 stars [8,9,15,18], and one study was rated 7/9 stars [17].
| Author, year | Study design | Sample size | Population characteristics: mean age, mean BMI, % smokers, % of family history of VTE | Name of COC | Event/person time – Event/number of participant a | Funding | Study quality (NOS scale) |
|---|---|---|---|---|---|---|---|
| Yonis, 2025 | Prospective study | Women* on E2-based COC: - n=14,135 Women* on synthetic estrogens: - n=2,092,749 |
Non-use of hormonal contraception: mean age: 32 mean BMI: 23 17.9% of smokers 8.1% of family history of thrombosisAny hormonal contraception use: mean age: 25 mean BMI: 23 16% of smokers 11% of family history of thrombosis |
E2-based COC: E2-DNG Synthetic estrogens: - EE/LNG - EE/NGM - EE/DSG - EE/GSD - EE/DRSP - EE/CPA - EE/DNG - EE/NET |
E2-based COC: - 7/14,135 Synthetic estrogens: - 997/2,092,749 |
Sygeforsikringen “Danmark” (grant 2021-0128). | 9/9 stars |
| Reed, 2021 | Prospective study | Women on E2-based COC: - n=44,559 Women on synthetic estrogens: - n=49,754 |
mean age: 30.1 mean BMI: 23.3 21% of smokers 2.4% of family history of VTE |
E2-based COC: - E2/NOMAC Synthetic estrogens: - EE/LNG |
E2-based COC: - 12/48,846 Synthetic estrogens: - 25/62,337 |
Merck Sharp and Dohme, a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA, and Theramex Ireland Limited, Dublin, Ireland | 9/9 stars |
| Schink, 2022 | Case-control study | Women on E2-based COC: - E2/DNG: n=35 - E2/NOMAC: n=66 Synthetic estrogens: - EE/LNG: n=2,512 |
VTE cases: mean age 17.6 20.6% obese 4.12% of smokers 10.6% of family history of VTEControls: mean age 17.6 6.67% obese 2.54% of smokers 1.43% of family history of VTE |
E2-based COC: - E2/DNG - E2/NOMAC Synthetic estrogens: - EE/LNG - EE/NGM - EE/DSG - EE/DRSP - EE/CMA - EE/CPA - EE/DNG - EE/NET - EE/GSD |
E2-based COC: - 6/101 Synthetic estrogens: - 744/7774 |
Funded by the Federal Institute for Drugs and Medical Devices (Bundesinstitut für Arzneimittel und Medizinprodukte, BfArM) | 9/9 stars |
| Heikinheimo, 2022 | Nested Case-control study | Women on E2-based COC: n=129 - E2/DNG: n=62 - E2/NOMAC: n=67Women on synthetic estrogens: - n=510 |
mean age: not reported mean BMI: not reported % of smokers: not reported % of family history of VTE: not reported |
E2-based COC: - E2/DNG - E2/NOMAC Synthetic estrogens: - EE/DSG - EE/GSD - EE/DRSP |
E2-based COC: - 25/129 Synthetic estrogens: - 158/510 |
Erkko Foundation; Yrjö Jahnsson foundation (ET); Avohoidon tutkimussäätiö & Helsinki University Library |
7/9 stars |
| Bauerfeind, 2024 | Prospective study | Women on E2-based COC: - n=11,616 Women on synthetic estrogens: - n=58,693 |
mean age 27.1 mean BMI 24.2 23% of smokers 3.1% of family history of VTE |
E2-based COC: - E2-DNG Synthetic estrogens: - EE-LNG - oCOC |
E2-based COC: - 11/17,932 Synthetic estrogens: - 99/107,586 |
Unconditional grant from Bayer AG, Germany | 9/9 stars |
Data are reported exclusively in women/years.
a Event/person time reported for prospective studies – Event/number of participants reported for case-control studies.
b Only confirmed VTE were considered as events.
Abbreviations: BMI: Body Mass Index; CMA: Chlormadinone Acetate; COC: Combined Oral Contraceptives; CPA: Cyproterone Acetate; DNG: Dienogest; DRSP: Drospirenone; DSG: Desogestrel; E2: Estradiol; E4: Estetrol; EE: Ethinyl Estradiol; GSD: Gestodene; LNG: Levonorgestrel; NET: Norethindrone; NGM: Norgestimate; NOMAC: Nomegestrol Acetate; NOS scale: Newcastle-Ottawa Scale; oCOC: Other Combined Oral Contraceptives (other than EE/LNG).
All studies were included in the EE-based versus E2-based model. Four studies contributed data to the EE/LNG versus E2-based model [8,9,15,18]. For this comparison, adjusted hazard ratios (HRs) were available for three studies [8,9,18].
Crude analyses
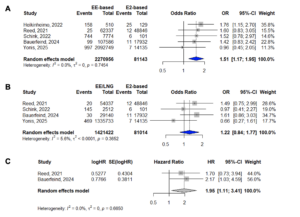
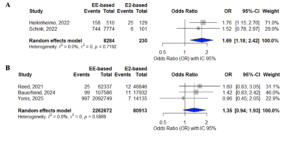
A significant 51% increase in VTE risk was observed among EE-based COC users compared with E2-based COC users (Peto OR 1.51; 95% CI 1.17–1.95) (Figure 2). The model showed no heterogeneity (I² = 0%, p = 0.7454). No significant association was found when comparing EE/LNG with E2-based COCs (Peto OR 1.22; 95% CI 0.84–1.77; I² = 5.6%, p = 0.3652) (Table 2).
| Type of analysis | Number of studies, sample size | Random effect model (crude Peto OR, [95% CI]) | Heterogeneity test (I², p for Q statistics) b | GRADE assessment a | |||||
|---|---|---|---|---|---|---|---|---|---|
| Risk of bias | Inconsistency | Indirectness | Imprecision | Publication bias | Level of Evidence | ||||
| EE-based COCs vs E2-based COCs | K=5 n EE-based = 2,275,520 n E2-based= 81,143 |
1.51 [1.17-1.95] | I² 0%, p=0.75 | Not serious | Not serious | Not serious | Not serious | Not serious | Moderate |
| EE/LNG vs E2-based COCs | K=4 n EE/LNG= 1,421,422 n E2-based= 81,014 |
1.22 [0.84-1.77] | I² 6%, p=0.37 | Not serious | Not serious | Not serious | Serious | Not serious | Low |
| Type of analysis | Number of studies, sample size | Random effect model (Adjusted HR, [95% CI]) c | Heterogeneity test (I², p for Q statistics) b | Risk of bias | Inconsistency | Indirectness | Imprecision | Publication bias | Level of Evidence |
| E2-based vs EE/LNG in studies with adjustment for confounding factors | k=3 n E2-based= 66,811 (E2/DNG) 66,834 (E2/NOMAC) n EE/LNG= 85,689 |
E2/DNG: 2.02 [1.18-3.48]
E2/NOMAC: 1.48 [0.80-2.72] |
E2/DNG: I2=0%, p=0.79E2/NOMAC: I2= 37%, p=0.20 |
Not serious | Not serious | Not serious | Not serious | Not serious | Moderate |
| Studies from ZEG E2-based vs EE/LNG |
k=2 n EE/LNG= 66,778 n oCOC=83,177 |
1.95 [1.11-3.41] | I2=0%, p=0.67 | Not serious | Not serious | Not serious | Not serious | Not serious | Low |
a GRADE assessment: 1) Study quality was considered as serious if low study quality was reported for more than 75% of the included studies, 2) Inconsistency was considered as serious in case of unexplained substantial significant heterogeneity, 3) Indirectness was considered serious if presence of factors that limit the generalizability of the results, 4) Imprecision was considered serious if k<3 or large 95% Confidence Intervals, 5) Publication bias : due to the low number of included studies (k=5 in the global meta-analytical model), a proper evaluation of publication bias could not be performed. However, because of the quality of the manual search performed for this systematic review and meta-analysis, any missing of evidence was considered as very unlikely and no serious publication bias was considered in the GRADE assessment.
b I2 is reported for adjusted HR when available, otherwise it is reported for crude Peto OR.
c Potential confounders considered in analyses: Reed et al.: age, body mass index, family history of VTE and current duration of HC use; Schink et al.: age at cohort entry, cardiovascular diseases, coagulation disorders and other blood diseases, diabetes or use of antidiabetics or insulin; migraine with aura, Varicose veins of lower extremities, obesity, Paresis, hospitalization, surgery, fractures or trauma, Current use of ASA, antiplatelets, antithrombotics or DOACs, Current use of NSAIDs, current use of glucocorticoids or other corticoids, Current use of antidepressants or antipsychotics; Bauerfeind et al.: age, body mass index (BMI), duration of current hormonal contraceptive use and family history of VTE; for ATE, it included age, BMI, smoking, treated hypertension and a family history of fatal ATE.
Adjusted analyses
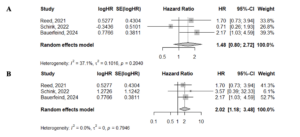
Adjusted HRs ranged from 1.48 (95% CI 0.80–2.72) for E2/NOMAC in Schink et al. to 2.02 (95% CI 1.18–3.48) for E2/DNG in Schink et al. (Supplementary Figures S1A and S1B). Schink et al. [18] reported two E2-based groups (E2/NOMAC and E2/DNG); therefore, analyses were run separately to avoid including the EE/LNG comparator arm twice. Restricting the analysis to the EMA-mandated PASS studies (k = 2) [8,9], the pooled adjusted HR showed a statistically significant increase in risk (HR 1.95; 95% CI 1.11–3.41) (Figure 2C).
Sensitivity analyses
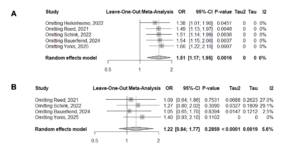
The leave-one-out analysis showed stable results in the EE-based versus E2-based model, with effect sizes ranging from 1.38 (95% CI 1.01–1.90) when omitting Heikinheimo et al. to 1.60 (95% CI 1.22–2.10) when omitting Yonis et al. (Figure 3). For the EE/LNG versus E2-based model, the leave-one-out analysis produced effect sizes ranging from 1.05 (95% CI 0.65–1.70) when omitting Bauerfeind et al. to 1.40 (95% CI 0.93–2.10) when omitting Yonis et al. (Figure 3).
Subgroup analyses by study design showed a significant 69% increase in VTE risk among EE-based COC users compared with E2-based COC users (Peto OR 1.69; 95% CI 1.18–2.42) when restricting evidence to case–control studies (k = 2) (Supplementary Figure S2). A non-significant 35% increase in VTE risk (Peto OR 1.35; 95% CI 0.94–1.93) was observed when restricting evidence to cohort studies (k = 3) (Supplementary Figure S2).
GRADE assessment
For the crude analyses, the level of evidence was rated as moderate for the EE-based versus E2-based model and low for the EE/LNG versus E2-based model. For the adjusted analyses, the level of evidence was considered moderate for the EE/LNG versus E2/DNG or E2/NOMAC models, and low for the EE/LNG versus E2-based model (i.e., the EMA-mandated PASS studies).
Discussion
This updated meta-analysis strengthens the evidence supporting body identical estrogens as a safer contraceptive option regarding VTE risk. We confirmed a significant 51% increase in VTE risk among EE-based COC users compared with E2-based COC users. No significant association was found for EE/LNG compared with E2-based COCs in the crude analysis. However, after adjustment for confounding factors, pooled HRs derived from the EMA-mandated PASS showed a significant 95% increase in VTE risk among EE/LNG users versus E2-based COC users. These findings emphasize the importance of accounting for confounding factors when evaluating VTE risk in users of body identical estrogen-based COCs.
VTE is a significant global public health concern, generating a substantial economic burden. The average societal cost over three years (2025-adjusted) for managing a single VTE event is estimated at €110,000 when considering lost productivity, hemorrhagic complications from anticoagulation, and VTE management itself [19]. When extrapolated to the current burden of COC-associated VTE, the economic impact may approach €2.5 billion annually in Europe alone [19,20]. Since the introduction of the first COCs, several strategies have sought to mitigate VTE risk [1]. Lowering the EE dose and considering progestin type have contributed to reductions in risk; however, incidence remains substantial, largely due to risk-mitigation strategies that do not sufficiently limit EE-based pill use in potentially high-risk populations [1,19]. Replacing EE with E2 or E4 may further reduce preventable VTE events [1] and offer an economic benefit to healthcare systems.
Multiple lines of evidence support the use of body identical estrogens. Biological studies have shown that these estrogens exert a lower impact on coagulation [6,21,22]. Higher nAPCsr values are observed with EE/LNG compared with COCs containing body identical estrogens [23-25]. A previous meta-analysis reported a significant 33% reduction in VTE risk with E2-based COCs compared with EE-based COCs, and after adjustment for confounding factors, a 49% lower risk among E2-based COC users compared with EE/LNG (HR 0.51, 95% CI 0.29–0.90; I²=0%, p=0.66) [10]. Consistent with that work, taking E2-based COCs as the reference showed no significant increase in VTE risk among EE/LNG users (peto OR 1.22, 95% CI 0.84–1.77), likely reflecting limited statistical power due to the low number of events.
Real-world pharmacovigilance data from the EMA’s EudraVigilance and the FDA’s FAERS databases align with these findings. A recent disproportionality analysis showed that E4/DRSP (PRR 0.24, 95% CI 0.17–0.33), E2/DNG (PRR 0.45, 95%CI 0.41–0.49), and E2/NOMAC (PRR 0.44, 95% CI 0.38–0.51) had significantly lower reporting of VTE events than EE/LNG, similar to progestin-only pills. FAERS analyses produced similar patterns, with E2/DNG (PRR 0.80, 95% CI 0.65–0.97) and E4/DRSP (PRR 0.54, 95%CI 0.30–0.99) showing the lowest reporting rates, comparable to DRSP (PRR 0.64, 95% CI 0.55–0.74) and NET-only pills (PRR 0.69, 95% CI 0.60–0.79) [26]. Beyond their safer thrombotic profile, body identical estrogens such as E4 also present more favorable interaction characteristics. They should be considered first-line options, particularly for women with multiple health risk factors [27-30].
Overall, these converging lines of evidence show that body identical estrogens combined with any progestin exhibit a more favorable VTE profile than EE-based COCs. Their use also facilitates progestogens such as drospirenone, which offer additional benefits including anti-mineralocorticoid and anti-androgen effects. As such, effective, well-tolerated, and safer body identical estrogen-based COCs can be used more broadly. With additional evidence, these formulations could become the new benchmark for COCs. As shown in this updated meta-analysis, when body identical estrogens are used as the reference standard, EE-based COCs—including EE/LNG—would likely trigger pharmacovigilance safety signals. Yet national and international guidelines still classify both EE-based and body identical estrogen-based COCs within the same risk category. Based on the previously reported 33% reduction in VTE risk with E2-based COCs [10], shifting first-line use to body identical estrogens could substantially reduce the annual number of VTE events. In Europe, a 33% reduction would lead to approximately 14,740 COC-associated VTE cases each year instead of 22,000, corresponding to potential annual savings of € 900 million [20].
Strengths and limitations of the model have been previously described [10]. This update has several strengths. First, it incorporates the large observational study by Yonis et al. [15]. Additional large-scale prospective studies assessing the safety of body identical estrogens remain necessary. Two regulator-mandated PASS—INAS-NEES and INAS-SEECS—have been developed to characterize and compare the safety of E4/DRSP, focusing on VTE events [31,32], with results expected after 2029. Second, the meta-analytical models were evaluated using the GRADE framework. Because randomized clinical trials are neither feasible nor ethical for assessing VTE risk with COCs, observational studies represent the only methodologically appropriate approach. In accordance with GRADE, we began at a moderate level of evidence. For EE-based versus E2-based associations, no downgrades were warranted, so this comparison should be interpreted as supported by a high level of evidence. In contrast, for the EE/LNG versus E2-based models (crude and EMA PASS), we downgraded due to imprecision, yielding a low level of evidence. However, considering the inherent constraints of this field, the evidence could reasonably be viewed as moderate.
Conclusion
These findings, grounded in biological, clinical, and real-world evidence, support a shift toward safer first-line contraceptive options. This updated meta-analysis reinforces the position of body identical estrogens as safer contraceptives with respect to VTE risk and suggests that formulations containing these estrogens should be prioritized over EE-based COCs such as EE/LNG.