Introduction
Placenta accreta spectrum (PAS) disorder is clinically diagnosed when the placenta cannot be separated from the uterus after delivery [1]. This abnormal attachment of the placenta to the myometrium occurs when a defect of the decidua basalis allows the chorionic villi to invade the myometrium [1].
Placental attachment disorders in which the placental villi extend into the uterine myometrium is the result of partial or complete absence of the decidua basalis and the Nitabuch’s layer (placenta accreta, approximately 81% of cases), extension deep into the myometrium (placenta increta, approximately 11% of cases) or the penetration of the entire myometrium (placenta percreta, 6%) and extension into the serosa, with some cases even infiltrating the bladder or rectum [2].
In patients with a prenatal diagnosis of PAS, the distinction between placenta accreta (International Federation of Gynaecology and Obstetrics [FIGO] grade 1), increta (FIGO grade 2) and percreta (FIGO grade 3) is of great importance when preparing for delivery. PAS disorder is responsible for adverse maternal outcomes at delivery, mainly due to severe postpartum hemorrhage, and remains the leading cause of peripartum hysterectomy in Western countries. Placenta percreta is associated with substantially increased maternal morbidity compared with placenta accreta/increta [1].
The incidence of the condition ranges from 1:533 to 1:2500 births, with a tenfold increase reported over the last 50 years [3,4]. The incidence of placental attachment disorders is increasing due to higher number of cesarean sections being performed (up to 67% after 3 or more cesarean sections) [5]. Miller et al. [6] demonstrated that women with placenta previa have a 9.3% incidence of placenta accreta, compared with a 0.005% incidence in women with normally located placentae.
Ultrasound (US) and magnetic resonance imaging (MRI) are the antenatal imaging modalities for the diagnosis of PAS. US is relatively inexpensive and is widely available. The cardinal findings of PAS on US include placental lacunae, loss of retroplacental clear space, reduction of myometrial thickness, and an irregular bladder wall with increased vascularity. MRI has been gaining attention as an alternative modality of choice for PAS evaluation and diagnosis especially in cases of posterior and lateral invasion, which are areas that are difficult to assess using US. MRI is also helpful for surgical planning since it accurately demonstrates the location of the placenta relative to adjacent structures (i.e., the cervix, bladder, and pelvic side walls) [7].
Placental attachment disorders are well known to cause significantly increased maternal and neonatal morbidity and mortality, and with its increasing occurrence, every obstetrician must be familiar with the most current diagnostic and treatment options [8]. However, limited data exist to guide the optimal management of this condition.
The aim of this study was to retrospectively analyze five patients diagnosed with placental attachment disorders who were treated in our training and research hospital.
Methods
This retrospective study included all 5 cases of placental attachment disorders treated in the Training and Research Hospital. Clinical information was obtained from the written delivery reports and databases of the Clinics of Obstetrics and Gynecology. PAS were suspected based on ultrasonography, followed by magnetic resonance imaging, verified during cesarean delivery, and confirmed by histopathological examination of surgical hysterectomy specimens.
Results
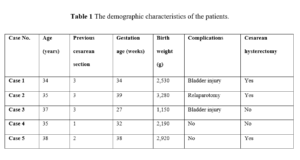
The demographic and clinical characteristics of the 5 women are reported in Table 1. Notably, for five women, four of whom had more than one cesarean delivery, PAS was diagnosed prenatally in five cases. Placental insertion was anterior and previa in all cases. The infiltration of the surrounding tissues and/or vesico-uterine fold was confirmed intraoperatively for all of the patients. Their placentas were left in situ after their fetuses had been extracted through a vertical fundal hysterotomy. Our patients received an average of 3 units of blood transfusion during and after the operation. One patient received 5 units of blood transfusion (2nd case).
Case 1
A 34-year-old pregnant woman with painful vaginal bleeding at 34 weeks of gestation who had a history of 3 previous cesarean section surgeries was admitted to the emergency room. Sonographic findings were protrusion of the placenta into the bladder, increased vascularity of the uterine serosa–bladder interface, placenta percreta was suspected in the transabdominal ultrasonography. She had undergone cesarean section immediately for continued vaginal bleeding at the clinical follow‐up. The midline incision was performed at the abdomen. Placenta caused a dilation and distention of the whole lower segment to approximately 10 cm below the fundus and serosa of the uterus causing increased vascularity. A vertical incision was performed below 6 cm of the fundus away from the placenta, and a 2,530 g healthy baby was delivered. Hysterectomy was decided because of the placental insertion anomaly. The lower segment of the placenta was fully occupied. The anterior uterine wall was then recognized to be affixed to the bladder. Dissection of the anterior uterine wall from the posterior bladder was accompanied. When the placenta was dissected, bladder injury occurred. Bladder injury was repaired. The patient was discharged and healthy on the 10th postoperative day.
Case 2
A 35-year-old pregnant woman at 39 weeks of gestation who was diagnosed with total placenta previa was admitted to the hospital. The patient had a history of 3 previous cesarean deliveries. The sonographic examination revealed placental lacunae, irregular placenta-myometrium interface (PMI) and hyperechoic uterus-bladder interface (bladder line). Preoperative evaluation of the patient suspected a PAS and delivered with cesarean section electively. When the midline incision was performed at the abdomen, a dilated area covering the cervix was observed. A transverse incision was performed at the upper segment of the uterus, and a 3,280-g healthy baby was delivered. A hysterectomy was decided to perform because of the lack of separation of the placenta. At the follow-up, after 5‐6 hours, the patient’s blood pressure dropped, and free intra-abdominal fluid was diagnosed at the US evaluation. A re-laparotomy procedure was performed. A total of 1,500 cc of blood and coagulum were viewed in the abdomen. Extensive bleeding was observed from collateral vessels emerging where the placenta invades the urinary bladder. Ligation of the internal iliac arteries was decided. She was discharged on the 8th postoperative day, healthy and with no clinical problems.
Case 3
A 37-year-old woman with three previous cesarean deliveries was referred due to vaginal bleeding at the 27th week of gestation according to US. Sonographic examination revealed the loss of the hypoechoic retroplacental space ("clear zone"), subplacental and utero-vesical hypervascularity and bridging vessels. Total placenta previa and PAS were suspected, hence, she was hospitalized for follow-up. Findings upon MRI scan included: thinning or loss of the retroplacental T2 dark zone, myometrial thinning, and placental bulge, irregular bladder wall, therefore PAS was suspected. The placenta implanted along the lower anterior uterine wall where it invaded beyond the uterus and involved portions of the bladder dome. Cesarean section was performed emergently because she had massive vaginal bleeding on the 15th day of hospitalization. Midline incision was performed. Placenta invading the bladder serosa was seen. A vertical incision was made in the uterus and a 1,150 g baby was delivered with the Bracht maneuver as a breech presentation. When the bladder was rejected, 2‐3 cm of bladder injury occurred. Hysterectomy was not performed. Bladder injury was repaired. She was discharged on the 8th postoperative day, healthy with no clinical problems.
Case 4
A 35-year-old woman who had one previous caesarian section was admitted with vaginal bleeding at the 32nd week of gestation. Placental lacunae, myometrial thinning an irregular border between the bladder and myometrium, and placenta previa was diagnosed on US examination. She was hospitalized for follow-up and had no active vaginal bleeding upon examination with a speculum. A marked heterogeneity of the placenta was detected in the MRI scan. This MRI examination was performed for preliminary diagnosis of the PAS. A midline incision was made in the abdomen, and a vertical incision was made in the uterus. A 2,190 g baby was delivered. The placenta invaded the serous layer of the bladder, and the lower segment of the uterus was tube‐like. Additionally, an increase in vascularization was observed. Bilateral hypogastric artery ligation was performed without needing a hysterectomy. Upon patient’s request, bilateral tubal ligation was performed with the Pomeroy technique. She was discharged on the 5th postoperative day, healthy and with no clinical problems.
Case 5
A 38-year-old pregnant woman, with a history of two previous cesarean sections, was admitted to the hospital at the 38th week of gestation. Utero-vesical hypervascularity and placental bulge towards the bladder were detected upon US examination, hence, total placenta previa and PAS were diagnosed. Midline incision was performed. The placenta caused dilatation of the lower segment of the uterus, also invading the bladder. A vertical incision was performed at the beginning of the fundus away from the placenta. A 2,920 g baby was delivered. Bilateral hypogastric artery ligation was performed. When the placenta was manually removed, the anterior wall of the uterus was perforated and the lower segment of the uterus was heavily damaged; that required hysterectomy due to massive vaginal bleeding. She was discharged on the 5th postoperative day, healthy and with no clinical problems.
Discussion
The present study indicates that maternal morbidity is significantly increased if pregnancy is complicated by PAS. PAS is a greatly feared obstetric complication, even when presenting at full-term delivery, as it may lead to emergency hysterectomy, massive hemorrhage, and urinary complications [9]. The risk for placenta accreta in a patient with placenta previa and prior cesarean delivery increases with the number of previous cesarean deliveries. Silver et al. [10] reported the risk of PAS for the first, second, third, fourth, and fifth or greater cesarean delivery to be 3.3%, 11%, 40%, 61%, and 67%, respectively [10]. It has been reported that the risk of requiring hysterectomy after normal vaginal labor was 1/30,000 and rose to 1/220 with two or more prior cesarean sections [11]. Three of our cases had had three previous cesarean sections.
Advanced maternal age is also a reported independent risk factor for placenta accreta, with the risk increasing every year beyond 20 years of age [4]. The mean age of our patients was 35.8 years. There are two major management options for PAS: cesarean hysterectomy and cesarean delivery with conservative management of the placenta. Hysterectomy has been for a long time the primary treatment for placenta accreta as proposed the ACOG, but it is associated with high morbidity and mortality, including the psychological consequences of uterine loss in young women [12]. The use of a midline vertical incision may be considered because it provides sufficient exposure if hysterectomy becomes necessary. A classic uterine incision, often transfundal, may be necessary to avoid the placenta and allow delivery of the infant. Generally, planned attempts at manual placental removal should be avoided. Placenta left in situ with methotrexate therapy is another form of care management. The 2010 French retrospective multicenter study included women over a 14-year period and reviewed 167 women treated with conservative management. Successful conservative treatment, defined as uterine preservation, was reported in 78%, but there was a 28% overall rate of infection, 11% incidence of delayed postpartum hemorrhage, and 6% occurrence of severe maternal morbidity, including sepsis, septic shock, and one maternal death (from complications secondary to methotrexate therapy) [13]. The complications associated with leaving the placenta in situ were primarily hemorrhage and infections, including a 58% risk of hysterectomy up to nine months after delivery [14].
After the operation of placenta percreta cases there will be a 25% possibility of needing reoperation, 77% needing intensive care unit support, 67% needing red blood cell transfusion excess the 10-units, 7% maternal mortality and 9% perinatal mortality [15]. None of our patients required intensive care unit. On average, our patients received 3 units of blood transfusion during and after the operation, with one patient receiving 5 units of blood transfusion (2nd case). Except for case 2, the other cases did not need surgical re-intervention. In one case (case 2), re-laparotomy was performed due to decreasing blood pressure and the US detection of intra-abdominal fluid. In this case bilateral hypogastric artery ligation was performed (case 2). Bilateral hypogastric artery ligation was performed during hysterectomy to control the hemorrhage in three of our cases (cases 2, 4, and 5). In our study, all 5 patients were discharged at an average of seven days postoperative (range 5‐10 days) and had no clinical problems upon follow-up.
The bladder is the most frequently involved extrauterine organ when there is a placenta percreta. Bladder involvement is associated with significant morbidity [16]. The incidence of concomitant bladder invasion is approximately 1 in 10,000 births [17]. Bladder invasion was detected in all of our cases. In women without overt invasion of the bladder at the time of surgery, an initial attempt to reflect the bladder may be carried out. Hemorrhage at the time of bladder reflection may be a sign of placenta percreta, and further dissection should be stopped. Leaving the placenta in situ would be a treatment option if the bladder could not be reflected or if the woman has the desire to conserve future fertility. When bladder reflection is carried out easily and there is no strong desire to maintain fertility, significant complications may be avoided if a hysterectomy is carried out at the time of cesarean section.
The diagnosis of adherent placenta is usually established by US and occasionally supplemented by MRI. The US features suggestive of placenta accreta include irregular shaped placental lacunae (vascular spaces) within the placenta, thinning of the myometrium overlying the placenta, loss of the retroplacental “clear space,” protrusion of the placenta into the bladder, increased vascularity of the uterine serosa–bladder interface, and turbulent blood flow through the lacunae on Doppler US [18]. The most valuable MRI features of placenta accreta include: dark intraplacental bands on T2-weighted sequences, uterine bulging, and heterogeneous signal intensity within the placenta [19].
All of our cases had preoperative diagnosis. Two patients who were admitted to our emergency service and hospitalized underwent MRI to support the diagnosis of insertion anomalies during preoperative follow-up. For cases 3 and 4, using both US and MRI findings we established the diagnosis of PAS. The US findings of PAS include the disappearance of the hypoechoic area between the urinary bladder and the placenta. These images were obtained in our cases. Preoperatively, in 2 cases by means of US and MRI we suspected anomalous placental invasion.
The delivery timing for placenta accreta must be individualized. Obstetricians must perform thorough counseling to include discussion of the high potential of performing a hysterectomy, profuse hemorrhage, probable transfusion, increased complications, and possible maternal death [20]. An expert opinion in 2010 recommended delivery for uncomplicated placenta previa at 36 to 37 weeks estimated gestational age and 34 to 35 weeks for suspected placental invasion [21]. In our cases, the reason for delivery before 35 weeks was bleeding or contractions. The reason for the operations performed at 38 and 39 weeks was that the patients did not have antenatal follow-up.
Conclusion
Antenatal diagnosis of a PAS, preoperative planning and a multidisciplinary approach may help save lives and reduce morbidity. The recommended management of suspected PAS is planned preterm cesarean hysterectomy with the placenta left in situ because attempts at removal of the placenta are associated with significant hemorrhagic morbidity, including hysterectomy, if necessary, as well as ensuring sufficient blood products. In addition, numerous unanswered questions exist regarding the optimal management of PAS. Prospective studies are warranted to decrease the morbidity of placenta accreta.