Introduction
Menopause, whether physiological or iatrogenic, is a life stage characterized by the permanent cessation of ovarian function and is associated with physiological, psychological, and cultural changes that influence sexuality in a biopsychosocial manner.
Female sexual interest and arousal disorder was first described in the diagnostic criteria of the DSM-5, in which, based on the available literature, the assessment of sexual desire and arousal disorders was combined into a single diagnosis [1]. However, many studies on the subject have used the DSM-IV classification, in which hypoactive sexual desire disorder (HSDD) was included within the category of sexual desire disorders and was defined by the absence or significant reduction of sexual thoughts or fantasies and/or desire for and receptivity to sexual activity, causing personal distress [2]. These indicators must persist over time (minimum of 6 months) and cannot be attributed to a mental illness, drug effects, other health problems, or relationship difficulties.
It is estimated that female sexual interest and arousal disorder (FSIAD) affects approximately 1 in 10 women of all ages and is the most prevalent female sexual dysfunction among postmenopausal women. Menopausal status has a significant impact on the prevalence of HSDD, with several studies showing higher rates in women who undergo surgical menopause at younger ages (16%–26%) compared with women in natural premenopause (7%–14%) [3]. In a European sample of women aged 50 to 70 years, the prevalence of HSDD was 9% in women with natural menopause and 12% in women with surgical menopause [2].
The proportion of women with low sexual desire increases with age, whereas the proportion experiencing distress related to low desire decreases over time. These data should be considered when proposing treatment, as a woman who does not report concern or distress about low desire would not meet the diagnostic criteria for FSIAD [2]. Low sexual desire is also associated with emotional or psychological distress, low self-esteem, and depression. Therefore, FSIAD leads to a significant deterioration in quality of life [2,3].
When FSIAD is present in postmenopausal women, after excluding other causes such as systemic diseases, mood disorders, and relationship problems, testosterone is an evidence-based therapy and is therefore included in clinical practice guidelines [2]. Despite this biological evidence, barriers to androgen therapy remain among both healthcare professionals and patients. This review analyzes the efficacy, safety, and clinical relevance of testosterone therapy (mainly in transdermal formulations) in postmenopausal women with baseline FSIAD, focusing primarily on randomized placebo-controlled clinical trials.
Methods
Literature search methodology
PubMed and MEDLINE were searched, along with a manual search, for studies published from 2000 to April 2025. To identify all relevant articles, the following search strategy was used: [("testosterone"[MeSH Terms] OR "testosterone"[All Fields]) AND ("menopause"[MeSH Terms] OR "menopause"[All Fields] OR "postmenopause"[MeSH Terms] OR "postmenopause"[All Fields]) AND ("sexual dysfunction, physiological"[MeSH Terms] OR "sexual dysfunction"[All Fields] OR "libido"[MeSH Terms] OR "libido"[All Fields] OR "sexual desire"[All Fields] OR "hypoactive sexual desire"[All Fields] OR "hypoactive sexual desire disorder"[All Fields] OR "sexual arousal disorder"[All Fields])]. This strategy was adapted and applied to different electronic databases.
Study selection
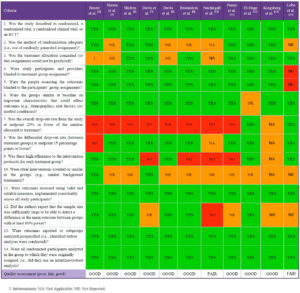
The publication time frame was limited including studies from 2000 to 2025. Meta-analyses, randomized controlled trials (RCTs) and systematic reviews were considered in this review. There was no language limitation and publications were included if the full text of the article was available. Articles based on animal models or evaluating other treatments for FSIAD were excluded as were those focusing on male sexuality. Studies were selected if the target population was postmenopausal women (with or without estrogen hormone therapy) and in natural or surgical menopause (with unilateral or bilateral oophorectomy), with FSIAD who were administered testosterone and compared with placebo or no treatment. Studies where the population was premenopausal women with FSIAD were excluded. The testosterone preparation used was in the form of a patch, gel or oral formulation. Studies using dehydroepiandrosterone (DHEA) were excluded, but not those including methyltestosterone. A reviewer (PTC) assessed trial eligibility and selected data from articles that may be most relevant to the treatment of FSIAD with testosterone in postmenopausal women, using a pre-specified protocol (information was collected on trial participant characteristics, type of intervention, how outcomes were measured and quality of studies) and, another reviewer (CCB) checked for any potential errors that may have occurred during data extraction, as outlined in Table 1.
The primary outcome measure was satisfactory sexual activities (SSA). Secondary outcomes were sexual desire (in most studies using the Profile of Female Sexual Function (PFSF)), distress associated with low sexual desire (mostly assessed with the Personal Distress Scale [PDS]), other domains of sexual function (arousal, orgasm, pleasure, sexual self-image), clinical relevance of treatment benefits and adverse effects (AE) in terms of safety and tolerability.
A total of 105 articles were reviewed –104 from the PubMed search and 1 from hand search– and 13 studies (12 from PubMed and 1 from hand search) were ultimately included in the synthesis [2]. The eligibility process is outlined in Figure 1 with a flow chart following the PRISMA statement.
Assessment of methodological quality and data extraction
The quality of studies included in the systematic review was assessed using the National Heart, Lung, and Blood Institute (NIH) Study Quality Assessment Tool to evaluate allocation, blinding of participants and investigators, and other sources of bias [4] (Table 2). The final assessment was reached by consensus between the two review authors (PTC and CCB).
Results
Literature search
The selection process is outlined in Figure 1. Of the 105 publications identified, 13 were selected for evaluation as they met the established inclusion criteria. The remaining 92 articles were excluded, mainly based on the title, but also due to study design and synthesis (Table 3).
Study characteristics
During the study period (2000–April 2025), a large number of studies evaluated the use of testosterone in postmenopausal women, either for the treatment of HSDD or for the assessment of changes in sexual desire in women without a diagnosis of sexual dysfunction. As shown in Table 1, two systematic reviews and 11 RCTs were included, with study populations ranging from 36 to 8,961 participants. Most studies demonstrated improvement in sexual function compared with placebo in postmenopausal women with HSDD; only one study specifically considered the diagnosis of FSIAD. Most studies analyzed transdermal testosterone at 300 μg/day (alone and/or in combination with estrogens +/- progestogens), although other routes of administration and doses were also evaluated.
The most widely used tool to assess sexual outcomes with testosterone treatment was the number of satisfactory sexual activities, although other validated questionnaires were also applied. The trials had a maximum follow-up of 24 weeks for efficacy and up to 4 years for safety.
Primary outcome measure: satisfactory sexual activity
Several studies have shown that transdermal testosterone at 300 µg/day improves satisfactory sexual activity (SSA) compared with placebo [2,3,5–12] (Table 1). In women with surgical menopause (WSM), the 300 µg/day testosterone patch significantly increased SSA, with a mean change of 1.56 activities per 4 weeks (corresponding to a 51% increase from baseline) compared with 0.73 activities per 4 weeks in the placebo group (p<0.001) [5] (Table 1). In this same study, significant differences were observed as early as week 5. Braunstein et al. [10] (Table 1), using a similar design in women with WSM, also reported a significant improvement from baseline of 0.58 SSA per week, representing a 79% increase, compared with a 43% increase in the placebo group (p=0.049).
Similarly, in women with natural menopause (WNM), administration of the 300 µg/day testosterone patch resulted in a mean change of 2.1 ± 0.28 versus 0.5 ± 0.23 SSA per 4 weeks compared with placebo (p<0.0001) [7] (Table 1).
In a study of postmenopausal women not receiving estrogen therapy, a significant increase in SSA frequency was reported in the group receiving 300 µg/day of testosterone compared with placebo (2.1 vs. 0.7 episodes per 4 weeks, p<0.001) [8] (Table 1).
One study [9] (Table 1) reported a non-significant increase in SSA frequency when comparing testosterone with placebo (p=0.06). However, analysis within the testosterone group alone showed a significant increase from baseline (p<0.0001), corresponding to a 148% improvement over baseline values.
In two studies [8,10], the 150 µg/day testosterone patch showed no significant improvement in SSA compared with placebo (1.2 vs. 0.7 episodes per 4 weeks, p=0.11) [8] (Table 1). The 450 µg/day testosterone patch produced results similar to those observed with the 300 µg/day dose, but with a higher incidence of adverse events [10] (Table 1). Treatment with a 1% testosterone cream (10 mg testosterone/day) resulted in a significant improvement in sexual function, as measured by the BISF-W, with a change of 8.76 versus 0.54 with placebo (p=0.0001) [13] (Table 1).
Secondary outcome measures
Sexual desire an associated distress
Most studies reported a significant increase in sexual desire and a significant decrease in distress associated with low desire. These domains were mainly assessed using the PFSF for sexual desire and the PDS for distress [3,5–7,9,12] (Table 1). Similar effects were observed at both testosterone patch dosages (150 μg and 300 μg). Davis et al. [8] (Table 1) showed a significant increase in sexual desire (PFSF) at both doses versus placebo (300 μg p<0.001; 150 μg p=0.04), along with a decrease in distress (PDS) (300 μg p<0.001; 150 μg p=0.04). In the study using 1% testosterone cream [13] (Table 1), no significant changes were observed in mood, depression, or stress, as assessed by the DASS (Depression, Anxiety and Stress Scale; p=0.52) and the POMS (Profile of Mood States; p=0.76).
It should also be noted that the combination of methyltestosterone plus estrogen significantly increased the SIQ (Sexual Interest Questionnaire) score compared with estrogen alone (2.8 ± 1.6 vs. 2.4 ± 1.4, p=0.047), with sexuality as a secondary outcome [14] (Table 1). Improvements were also observed in other domains of sexual function, including arousal, orgasm, pleasure, self-image, and reduced worry [5,9] (Table 1).
Clinical relevance of treatment benefits
Efficacy data are less meaningful without corresponding clinical relevance. In this regard, one study [15] (Table 1) showed that a significantly higher proportion of women in the testosterone group reported a meaningful overall benefit compared with those receiving placebo (33 of 64 women (52%) in the 300 μg/day testosterone patch group vs. 21 of 68 women (31%) in the placebo group; p=0.025). Women receiving testosterone were also significantly more likely to report that the treatment met their expectations than those receiving placebo (30 of 64 women (47%) vs. 18 of 67 women (27%); p=0.03).
Women who reported a benefit were significantly more likely to express willingness to continue testosterone treatment [15] (Table 1). More than 85% of women who experienced a significant benefit reported they were probably or definitely interested in continuing treatment, whereas more than 90% of women who did not experience a significant benefit reported they were probably or definitely not interested in continuing treatment [15] (Table 1).
Adverse effects impacting safety and tolerability
The incidence of androgenic adverse effects (acne and hair growth), mostly mild, was significantly higher in the testosterone group compared with placebo, but there were no significant differences in changes in facial hair [7,9,12,15]. No differences were observed between groups in alopecia or voice deepening [15]. Overall, there was no significant difference in the total number of adverse effects or severe adverse events.
Lipid profile, carbohydrate metabolism, cardiometabolic markers, and renal and hepatic function were not adversely affected. However, recommendations do not extend to women with cardiovascular disease, as they were not included in the clinical trials [2].
In one study [8] (Table 1), breast cancer was detected in three women assigned to the testosterone group (one of whom later reported symptoms before randomization, and one of the other two was diagnosed only after four months of therapy). An additional case of breast cancer was reported in one of the testosterone groups at the end of the study extension. The study authors noted that the excess cases of breast cancer in women treated with testosterone could be due to chance; however, they also suggested that a possible causal relationship should be considered. Some epidemiological studies have shown an association between endogenous or exogenous testosterone and breast cancer risk, while others have not. The article [8] also noted that some data suggest that adding testosterone to estrogen plus progestogen regimens may enhance hormonal stimulation of the breast, and that long-term data from large clinical trials on testosterone use are lacking.
One article [16] reported abnormal uterine bleeding, but no increased risk of endometrial hyperplasia or cancer was detected (biopsy showed atrophy or insufficient tissue). Another article [13] reported the development of virilization after a minimum of six months of treatment with intramuscular testosterone and also described clitoromegaly associated with long-term use of injectable androgen and estrogen therapy.
Discussion
For many years, testosterone has been prescribed to women to relieve a variety of symptoms, although its true risks and benefits remained uncertain [2]. Over the last two decades, however, researchers have devoted significant effort to clarifying its safety and therapeutic value. Transdermal delivery systems—namely patches and gels—offer noninvasive alternatives to injections, avoiding first-pass hepatic metabolism and enabling more physiological delivery through direct skin absorption. However, the two modalities differ in absorption kinetics, variability, ease of use, side-effect profiles, and practical clinical considerations [17]. The patch produces a mean testosterone profile that more closely mimics the circadian rhythm, whereas the gel appears to have greater inter- and intra-subject variability [18]. Both gels and patches effectively raise serum testosterone into the physiological range and improve symptoms of hypogonadism (low libido, mood changes, and loss of muscle mass) [19]. Because patches more closely mimic circadian rhythms while gels produce a flatter profile, some clinicians may prefer the patch when circadian hormonal patterns are a key consideration; conversely, gels may provide more flexibility in dosing and application site [20].
Current evidence suggests that systemic transdermal testosterone, administered at a physiological premenopausal dose (300 μg/day), provides a moderate improvement compared with placebo in postmenopausal women with HSDD unrelated to modifiable causes or comorbidities [21]. Importantly, no severe short-term adverse effects have been documented. Combined approaches that also address psychological and sociocultural factors remain valuable [22]. If testosterone therapy is pursued, counseling should be provided within a biopsychosocial model of sexuality, framing testosterone not as a cure but as a pharmacological aid within a broader, complex process.
Although multiple reviews have addressed testosterone therapy in postmenopausal women, this review emphasizes study populations and measurement tools, underscoring the need for more rigorously designed trials. Only three studies [5,6,12] used psychometric instruments that meaningfully assess female sexual desire and enrolled appropriate participants (postmenopausal women with diagnosed sexual dysfunction). Even with this stricter lens, the findings remain aligned with current scientific society recommendations for systemic transdermal testosterone in postmenopausal women with FSIAD. Still, further well-designed studies are necessary.
There is consensus among multiple international societies recommending testosterone therapy for postmenopausal women with decreased sexual interest (with or without reduced arousal) that causes personal or relational distress and who seek treatment.
Based on the studies evaluated in this review, testosterone therapy—mostly assessed in transdermal formulations at a dose of 300 μg/day—has shown statistically significant and consistent improvements in postmenopausal women with FSIAD across several domains of sexual function. These include the frequency of satisfactory sexual activities, sexual desire, orgasm, pleasure, sexual self-image, and personal distress associated with low sexual desire, in both women with natural and surgical menopause, with or without concomitant estrogen +/- progestogen therapy.
Testosterone therapy is generally well tolerated, with the most frequent adverse effects being mild and androgenic in nature (i.e. acne and hair growth). No significant differences were observed between testosterone-treated groups and placebo in either the total number of adverse effects or the incidence of severe adverse events. Long-term safety data (up to four years) generally show no major adverse events in laboratory parameters.
Patient selection is important. For women with sensitive skin or frequent irritation, gels may be preferable. For those who value a “set-and-forget” device and do not have issues with patch adhesion, a patch may offer more physiological circadian mimicry. Counseling is critical when using gels: patients must understand the risk of transfer to others, the need to apply the product to dry, intact skin, allow it to dry before dressing, and avoid contact with others for a defined period.
Overall, this review consolidates key information on the utility of testosterone in treating female sexual interest and arousal disorder in postmenopausal women, emphasizing its established efficacy and generally favorable safety profile. However, data remain limited regarding well-being, bone health, cognition, menopausal symptoms, mood disorders, breast cancer, cardiovascular disease, and long-term safety.
Author contributions
Patrícia Torné Castells and Camil Castelo-Branco contributed to the elaboration of this study, revising and approving final version to be submitted