Introduction
Adolescents frequently report menstrual-related symptoms that impact daily functioning and well-being, with combined oral contraceptives (COCs) often prescribed for symptom management and cycle regulation [1]. Several studies indicate that 70–90% of adolescents experience menstrual pain, and around one-third report symptoms severe enough to interfere with school attendance, social activities, or mood [2-5]. Despite this high prevalence, menstrual symptoms are often normalised and undertreated, underscoring the importance of assessing their broader effects on quality of life [2,6].
Despite their widespread use, data on the broader impact of COCs on adolescent health-related quality of life (HRQoL) remain limited. In particular, age-appropriate patient-reported outcomes (PROs) are underutilised in clinical research, despite their importance in capturing the adolescent perspective directly. Instruments such as the KIDSCREEN-27 offer a structured and validated approach to evaluating multidimensional aspects of well-being in this population [7,8].
Estetrol (E4), a native estrogen with favourable haemostatic and metabolic profiles, combined with drospirenone (DRSP), forms a modern COC option with a 24/4 regimen shown to be well-tolerated in adults [9-13]. The MIT-Es001-C303 (EudraCT 2019-003002-27, NCT04696511) study was the first trial to evaluate E4/DRSP in adolescents 12 to 17 years old. The study was conducted in accordance with Paediatric Investigation Plan (PIP) agreed upon with the European Medicines Agency (EMA). The primary safety and tolerability outcomes of this Phase 3 study have been reported separately and provide detailed data on adverse events, bleeding patterns, and dysmenorrhea outcomes [14]. Here, we present additional pre-specified PRO outcomes from the same study, specifically assessing the effects of E4/DRSP on HRQoL and bothersome menstrual symptoms.
Methods
We conducted this phase 3, multicentre, open-label, single-arm study between December 2020 and November 2023 across six European countries: Estonia, Finland, Georgia, Latvia, Poland, and Sweden. The study enrolled postmenarchal female adolescents aged 12 to 17 years and 2 months inclusive, with a menstrual cycle length of 21-45 days, who received 15 mg E4 in monohydrate form combined with 3 mg DRSP in a 24/4 regimen for six consecutive cycles. No minimum duration since menarche was required. After screening, eligible participants entered a pre-treatment (baseline) cycle. Participants who were already using a hormonal contraceptive stopped this method at the end of their regular cycle and entered the pre-treatment cycle without using hormonal contraception. Concomitant medication use was managed in accordance with the study protocol and the Summary of Product Characteristics (SmPC) for E4/DRSP. Use of other hormonal contraceptives or interacting drugs was not permitted, while non-hormonal treatments judged not to affect efficacy or safety were allowed. Condoms were provided to all participants for the duration of the study, if needed.
Eligibility was defined according to the product label [15]. Key exclusions included contraindications to COC use, abnormal vaginal bleeding, clinically relevant infections or disorders (e.g. pelvic inflammatory disease, lower genital tract infection), malignancy, use of long-acting contraceptives, or medications known to interact with the study treatment.
The study team conducted all procedures in accordance with the Declaration of Helsinki, Good Clinical Practice, and a Paediatric Investigation Plan approved by the European Medicines Agency. Ethics Committees at each site approved the protocol, and all participants and/or their legal representatives provided written assent or consent. It was approved by the local Ethics Committees at the respective centres: in Estonia by the Research Ethics Committee of the National Institute for Health Development (NIHD REC, No 533, 25 November 2020); in Finland by the Hospital District of Helsinki and Uusimaa Ethics Committee (Record No. HUS/1546/2020, 04 November 2020); in Georgia by the Independent Ethics Committee of LLC (No 22/05-03, 29 April 2022); in Latvia by the Independent Ethics Committee for Clinical Trials of Medicinal Products (No 201120-213/689a, 20 November 2020); in Poland by the Bioethics Committee at the Regional Medical Chamber in Białystok (No 41/2022/VIII, 18 May 2022); and in Sweden by the Swedish Ethical Review Authority (No Dnr 2020-06373, 15 December 2020).
Measurements
All PROs were collected using electronic diaries (e-diaries) completed in local languages using validated translations. Participants entered data at baseline, during cycles 1, 3, and 6, and at early termination. The e-diary included two validated instruments: the Menstrual Distress Questionnaire–Short Form (MDQ) to evaluate menstrual symptoms and the KIDSCREEN-27 for assessing HRQoL [7,16-18].
The MDQ is a self-reported validated instrument for measuring cyclical premenstrual (4 days before menstrual flow) and menstrual (first day of bleeding) and intermenstrual symptoms and comprises 46 items clustered in 8 domains: ‘Pain’, ‘Water Retention’, ‘Negative Affect’, ‘Impaired Concentration’, ‘Autonomic Reactions’, ‘Behaviour Change’, ‘Arousal’, and ‘Control’ [17,18]. Symptoms are scored using a 5-point severity scale ranging from 0 (no experience of symptoms) to 4 (symptoms are present, severe).
The KIDSCREEN-27 is a self-reported questionnaire developed for children and adolescents aged 8–18 years. It was developed through the European KIDSCREEN Project and validated across multiple countries and languages, ensuring cultural and linguistic equivalence [7]. It includes 27 items across five dimensions: Physical Well-being, Autonomy & Parent Relations, Psychological Well-being, Social Support & Peers, and School Environment. These dimensions capture a broad spectrum of adolescent experiences, including physical activity and energy levels, perceived independence and family support, emotional balance and self-esteem, the quality of peer relationships, and aspects of school functioning such as concentration and interactions with teachers. Each item is rated on a 5-point Likert scale, reflecting either the frequency (from "never" to "always") or intensity (from "not at all" to "extremely") of experiences during the past week.
Participants completed both the MDQ Form C (Cycle) and the KIDSCREEN-27 once per cycle between Day 7 and Day 14, at baseline and during Cycles 1, 3, and 6. The MDQ captured symptom severity across the menstrual, premenstrual, and intermenstrual phases within the previous cycle, whereas the KIDSCREEN-27 assessed general health-related quality of life over the previous week. Adherence recorded in the e-diary was verified by comparing electronic entries with returned tablet counts at each visit [14].
Analysis
We present the MDQ results for the four domains related to menstrual symptoms previously demonstrated to be the most bothersome [19]: the physical domains of ‘Pain’ (6 items) and ‘Water Retention’ (4 items) and the emotional domains of ‘Negative Affect’ (8 items) and ‘Impaired Concentration’ (8 items). The domains ‘Behaviour Change,’ ‘Autonomic Reactions’, ‘Arousal’, and ‘Control’ contain questions unrelated to the most bothersome menstrual symptoms and were not included in the analysis. Because bothersome symptoms are typically not prevalent during the intermenstrual phase [17,18], we only evaluated outcomes in the premenstrual and menstrual phases. We excluded a domain score for an individual participant if the score for more than two domain items was missing. When a score for one domain item was missing, we calculated the mean score of the remaining items and added it to the total raw score [18]. Domain raw scores (sum of item scores within each domain) were converted to t-scores using the conversion tables from the MDQ-C manual. This conversion allows comparison of MDQ results between cycles, across cycle phases, and between participants [18].
Raw KIDSCREEN scores for each domain were transformed into t-scores, as provided by the KIDSCREEN developers, to enable direct comparisons with international reference data [7,20]. Normative values for interpreting t-scores were derived according to the KIDSCREEN manual, using the reference group mean ± 0.5 SD as the range for ‘normal’ in line with the KIDSCREEN manual. This range reflects a clinically meaningful difference from the European reference population and supports identifying individuals with noticeably lower or higher HRQoL [16].
We included all participants who received at least one dose of E4/DRSP and completed both baseline and Cycle 6 questionnaires. All data from baseline, Cycles 1, 3, and 6 were summarised using means and standard deviations (SD). Changes in KIDSCREEN-27 and MDQ t-scores from baseline to Cycle 6 were assessed using the Wilcoxon signed-rank test for non-parametric comparisons. Both the MDQ and KIDSCREEN-27 comprise several distinct domains and do not yield a total score. Because these instruments measure conceptually separate constructs, no formal multiple-testing correction (e.g. Bonferroni or Holm) was applied. A p-value < 0.05 was considered statistically significant. Statistical analyses were performed using the SAS System for Windows, version 9.4 (SAS Institute Inc., Cary, NC, USA). The study aimed to include ≥100 participants, a number considered adequate to evaluate the safety of a new COC in adolescents, the primary objective of the study, and aligned with the requirements of the EMA-approved PIP. Therefore, no formal power analysis was performed.
In addition to analysing mean MDQ t-scores, two complementary analyses were conducted to provide further insight into changes in symptom severity. First, we calculated the overall distribution of participants across the five MDQ symptom severity categories (none, mild, moderate, strong, and severe) at baseline and at Cycle 6, using the total analysis population as the denominator. This group-level analysis describes how symptom severity is distributed within the entire analysis population at each time point but does not provide details on shifts between severity categories. For example, an increase in the proportion of participants with “no symptoms” at Cycle 6 may reflect improvements from various baseline categories (i.e., mild, moderate, strong, or severe). However, these transitions are not captured in this type of analysis.
To explore these transitions in more detail, we conducted a shift analysis. For each symptom, we calculated the proportion of participants within each baseline severity category who remained in the same category or moved to another severity level by Cycle 6. This approach provided a more detailed view of symptom evolution by capturing both the direction and extent of change from baseline.
We generated shift figures for symptoms that met both criteria: (1) at least 10% of the analysis population reported strong or severe symptoms at baseline, and (2) at least 40% of those participants showed a change in severity from baseline to Cycle 6. In the results section of this report, we included the data and shift figures for two representative symptoms (Cramps and Mood Swings); additional shift figures are provided as supplemental. A ≥20% difference between the proportions improving versus worsening was considered a clinically meaningful change.
Results
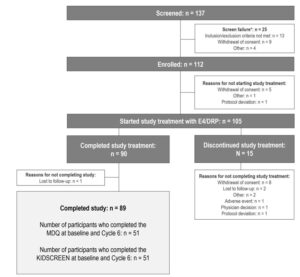
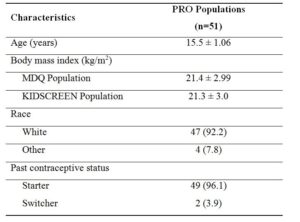
Of the 112 enrolled adolescents, 105 received at least one dose of the study drug, and 89 of them completed all six treatment cycles. Across all cycles, over 71% of participants adhered fully to the tablet intake (both active and placebo). Adherence to active tablets increased from 77.8% in Cycle 1 to 94.9% in Cycle 6 [14]. A total of 51 participants completed the MDQ and the KIDSCREEN questionnaires at baseline and Cycle 6 (Figure 1). Demographics and characteristics are presented in Table 1.
Data are presented as n (%) or mean ± standard deviation. MDQ: menstrual distress questionnaire. PRO: patient-reported outcome.
MDQ t-scores
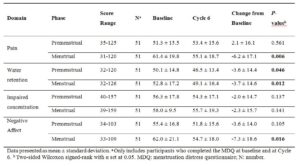
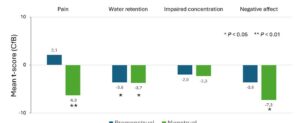
Across the four domains, t-scores decreased (except for premenstrual Pain), with more pronounced changes in the menstrual phase, particularly for Pain and Negative Affect (Figure 2). Improvements were observed for menstrual Pain, Water Retention, and Negative Affect, as well as for premenstrual and menstrual Water Retention (all p < 0.05) (Table 2, Figure 2).
MDQ-Shift Analysis
At baseline, at least 10% of participants reported strong or severe symptoms for 21 of the 26 domain symptoms. Of these, 21 symptoms also met the additional criterion of ≥40% of participants experiencing a change in severity from baseline to Cycle 6 (Supplementary Table S1).
Within this group of symptoms, participants reported notable improvements (>20% difference between proportions improving versus worsening) for menstrual cramps, backache, and general aches and pains (Pain domain); premenstrual and menstrual painful or tender breasts (Water Retention domain); and menstrual mood swings, crying, tension, and feeling sad or blue (Negative Affect domain) (Table 3).
In the physical and emotional domains, Pain and Negative Affect showed the largest t-score reductions during the menstrual phase (–6.2 and –7.3, respectively; Table 2), with 52.9% of participants experiencing improvement in menstrual cramps and 45.1% in menstrual mood swings (Table 3). Shift analyses for these symptoms are shown in Figures 3 and 4, respectively, with additional supplementary figures S1 through S12 for other symptoms. Overall, shifts were more pronounced for those reporting improvement compared to those reporting worsening.
Complete data for all 26 symptoms are presented in Supplementary Table S1.
KIDSCREEN Scores
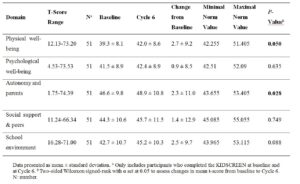
Numerical increases were observed from baseline to Cycle 6 across all KIDSCREEN-27 subscales, with statistically significant improvements in Physical Well-being and Autonomy & Parent Relations (p < 0.05) (Table 4).
At baseline, mean t-scores ranged from 39.3 (Physical Well-being) to 46.6 (Autonomy & Parent Relations), with all domains except Autonomy & Parent Relations below the corresponding reference means (± 0.5 SD). By Cycle 6, Physical Well-being, Psychological Well-being, Social Support & Peers, and School Environment scores had moved into the normal range (Table 4).
Discussion
In this study, we assessed menstrual symptoms and health-related quality of life in postmenarchal, treatment-seeking adolescents using a novel COC containing the natural estrogen E4 and the progestin DRSP. We observed an improvement in menstrual symptom severity and a modest but consistent improvement within the normal range in HRQoL over six treatment cycles using two validated PROs, the MDQ for menstrual symptoms and the KIDSCREEN-27 for broader HRQoL. MDQ results showed that E4/DRSP reduced the severity of menstrual symptoms, particularly during the menstrual phase.
T-scores fell significantly in the Pain, Water Retention, and Negative Affect domains, indicating a clear drop in symptom burden after six cycles. In the premenstrual phase, only Water Retention improved significantly. Overall, E4/DRSP alleviated symptoms across both cycle phases, with the most significant benefit observed during menstruation, when symptoms typically peak.
In the European phase 3 adult trial, scores for Pain, Water Retention, and Negative Affect dropped by –1.4 to –3.5 points. In our adolescent cohort, reductions ranged from –3.6 to –7.3 points, suggesting equal or greater benefit. As in the adult trial, we saw no apparent change in Impaired Concentration. This aligns with Boyle’s factor structure analyses, which found that Impaired Concentration is not an independent MDQ domain in healthy young women unless symptoms are more severe [21].
Shift analysis revealed that most adolescents improved to milder categories, often to no symptoms, with minimal movement in the opposite direction. The most significant shifts occurred for cramps, backache, general aches and pains, breast tenderness, mood swings, crying, tension, and feeling sad or blue.
This is the first study to apply the KIDSCREEN-27 to evaluate HRQoL in the context of contraceptive use. At baseline, t-scores in most domains were below European normative means, especially in Physical Well-being and Psychological Well-being. Following treatment, improvements were observed across all domains, with significant gains in Physical Well-being and Autonomy & Parent Relations. Although changes did not exceed the 5-point threshold for clinical significance [7,20], the short study duration and the cyclical nature of symptoms make these gains noteworthy. Importantly, scores for Physical Well-being, Psychological Well-being, Social Support & Peers, and School Environment moved from below-normative to within the range by Cycle 6, indicating a shift toward age- and sex-appropriate perceived well-being.
Although mean KIDSCREEN-27 changes did not reach the conventional 5-point threshold, several domains moved from below-normative to within-normative ranges by Cycle 6. This shift toward normative values is clinically interpretable, even when mean changes are modest, as it reflects a restoration of perceived well-being consistent with age- and sex-matched peers. Such normalisation provides meaningful context for interpreting HRQoL improvements in adolescents, where smaller numerical changes can still reflect substantial perceived benefit.
The MDQ detected larger and more distinct improvements than the KIDSCREEN-27 because it focuses on specific menstrual symptoms, including emotional items such as mood swings, crying, and tension, which showed marked improvement. The KIDSCREEN’s broader HRQoL scope may dilute changes directly linked to symptom relief. However, it has the significant advantage of providing normative reference data, allowing a clear clinical interpretation of whether scores fall within or outside the expected range for age- and sex-matched peers. This makes even modest changes meaningful, as shown by the shift of Physical Well-being, Psychological Well-being, and School Environment from below normative to normative levels by Cycle 6.
For the MDQ, no validated minimal important difference has been established. We therefore interpret changes based on domain-level trajectories and responder perspectives. Most adolescents shifted toward milder categories—often to no symptoms—and several symptom domains showed ≥20% net improvement. These domain-level and shift analyses provide converging evidence of clinically relevant improvement, even in the absence of an established minimal important difference.
Collecting PROs in adolescents is increasingly recognised as a key component of evaluating treatment impact and overall well-being. However, methodological challenges persist, including limited validation of available instruments, developmental differences in self-reporting, and the difficulty of maintaining consistent engagement over time. Recent systematic reviews of PRO instruments in children and adolescents emphasise the need for validated, multidimensional tools and highlight the practical challenges associated with sustained participation and accurate recall [8]. Our study demonstrates the feasibility of collecting detailed PRO data from adolescents using validated electronic diaries, although completion rates were suboptimal. This highlights the challenges of sustained self-reporting in this age group. Nevertheless, we collected sufficient data to reveal consistent and clinically interpretable patterns across two complementary, validated PRO tools, one symptom-focused and one broad HRQoL measure. The use of these instruments, together with extensive analyses including shift analyses and the use of normative data, is a strength of this study. Limitations include the single-arm, open-label design, the relatively small number of adolescents completing all PRO assessments, and the relatively short study duration. No adjustment for multiple testing was applied as both the MDQ and KIDSCREEN assess multiple, distinct constructs rather than repeated measures of a single outcome. Potentially relevant domains such as sleep, fatigue, and academic performance were not assessed.
Although this study suggests favourable trends in menstrual symptom relief and improvements in HRQoL and mood with E4/DRSP, these findings must be interpreted with caution due to the mentioned limitations and their exploratory nature. In addition, the results may partly reflect placebo or expectancy effects. The results are consistent with the adult Phase 3 E4/DRSP study, where similar MDQ patterns were observed [22]. Nevertheless, larger studies or post-marketing clinical experience will be valuable to confirm these outcomes.
Although the importance of comprehensive contraceptive counselling is well recognised, we wish to emphasise its central role in supporting method selection and continuation in adolescents. Structured, age-sensitive counselling should engage adolescents in shared decision-making and prepare them for expected bleeding patterns, potential side effects, and inform them about non-contraceptive benefits. In the authors’ view, this is especially critical in today’s digital environment, where adolescents often form opinions on contraception via social media platforms. Influencer narratives and peer testimonials, both accurate and inaccurate, can powerfully shape perceptions. By proactively addressing misinformation, particularly regarding mood changes and weight gain, clinicians can help adolescents make informed, confident choices.
Growing attention has been directed to the environmental impact of estrogens used in hormonal contraception, a topic that is also increasingly important to young people concerned about sustainability. Synthetic compounds such as ethinylestradiol (EE) are highly persistent and potent in aquatic ecosystems [23,24]. This creates a paradox: although effective contraception reduces environmental exposure to natural estrogens from pregnancies, the synthetic estrogens used in most oral contraceptives contribute their own persistent environmental load, reinforcing the importance of developing formulations based on estrogens with lower ecological impact. Environmental sustainability is also increasingly important to young people. Relative to other estrogens like EE, E4 is expected to be less persistent in the environment and has a lower potential for bioaccumulation in organisms. Ecotoxicology studies also show that E4 is less toxic to fish than EE and therefore suggest that E4 presents a more favourable environmental profile [25-27]. Combined with its profile as a natural estrogen-containing COC, E4/DRSP aligns with emerging preferences for “body-identical” hormones, as both E4, a native estrogen, and drospirenone, which shares pharmacological similarities with endogenous progesterone, provide hormones closer to those the body naturally produces [28]. Highlighting this combination of eco-friendly properties, physiological hormone profiles, and clinical benefits may enhance the appeal of E4/DRSP for adolescents who value both sustainability and well-being in their contraceptive choices.
Conclusions
E4/DRSP use over six cycles was associated with substantial reductions in menstrual symptom severity and modest but consistent improvements in HRQoL in postmenarchal, treatment-seeking adolescents. Importantly, KIDSCREEN-27 scores for Physical Well-being, Psychological Well-being, Social Support & Peers, and School Environment shifted from below-normative to normative levels, indicating a restoration of age- and sex-appropriate well-being. These findings support E4/DRSP as a promising option for adolescents with bothersome menstrual symptoms and demonstrate the value of integrating both symptom-specific and HRQoL measures in contraceptive trials. Given the study limitations, longer-term and controlled studies are warranted to confirm these benefits and explore their broader impact on adolescent health and daily functioning.
Author contributions
Conceptualization, All authors; Methodology, All authors; Formal Analysis, NF; Investigation, LP, ALH, KGD, KH, ND, GC and DA; Resources, LP, ALH, KGD, KH, ND, GC and DA; Data Curation, NF; Writing – Original Draft Preparation, NF; Writing – Review & Editing, All authors; Visualization, NF; Supervision, All authors. All authors have reviewed and agreed to the final version of the manuscript.